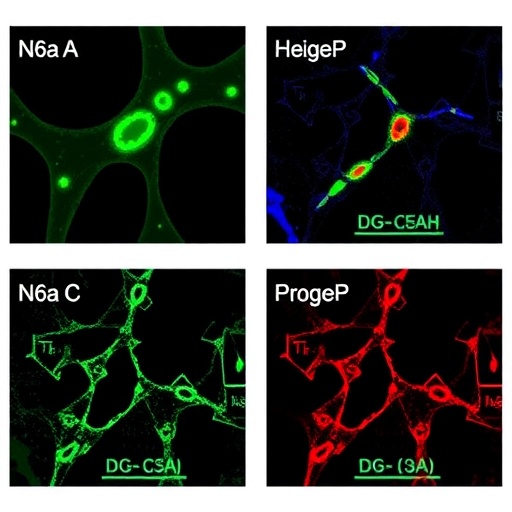
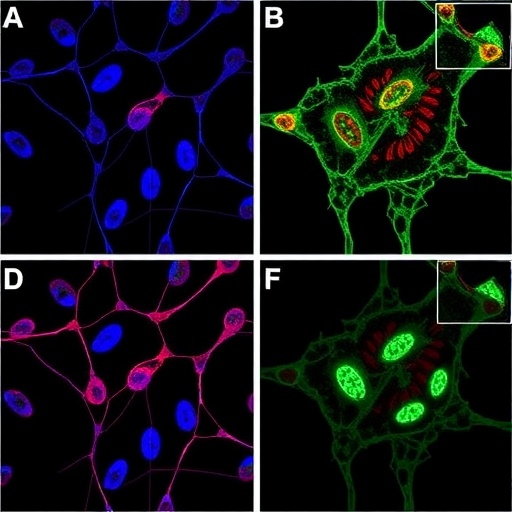
Recent research has made significant strides in unveiling the molecular underpinnings of osteosarcoma, a challenging and aggressive bone malignancy predominantly affecting children and adolescents. A pivotal study led by Chen et al. investigates the role of METTL3, a pivotal methyltransferase, in the dynamic landscape of m^6A modification, particularly its influence on the CACNA1E gene. This research not only sheds light on the intricate regulatory mechanisms driving osteosarcoma progression but also highlights potential therapeutic targets for addressing chemoresistance—a significant hurdle in effective cancer treatment.
The growing body of evidence underscores the critical function of m^6A methylation in various biological processes, particularly in cancer biology. METTL3, as an m^6A methyltransferase, catalyzes the addition of a methyl group to the adenosine residues of messenger RNA, ultimately influencing the stability, splicing, and translation of RNA molecules. In the context of osteosarcoma, the findings from Chen et al. suggest that METTL3-mediated modification of CACNA1E serves as a driving force in the oncogenic pathways promoting tumor growth and therapy resistance.
In this intricate web of cellular signaling, WNT signaling emerges as a crucial player. Chen et al. elucidate the mechanism by which CACNA1E, a gene encoding a calcium channel, is subjected to m^6A modification by METTL3. The study reveals that this modification enhances the expression of WNT7B, a member of the WNT family implicated in critical processes such as cell proliferation and differentiation. By augmenting WNT7B-mediated calcium signaling, METTL3 not only facilitates osteosarcoma cell proliferation but also contributes to chemoresistance, complicating treatment regimens for affected patients.
The link between calcium signaling and cancer progression is particularly compelling. Elevated intracellular Ca^2+ levels have been shown to influence various signaling cascades inherently tied to cellular growth and survival. As the study highlights, the interplay between METTL3, CACNA1E, and WNT7B creates a feedback loop that exacerbates tumor characteristics. This novel understanding paves the way for targeted interventions that could disrupt these oncogenic processes, offering hope to patients battling resistant forms of osteosarcoma.
A notable aspect of this research is its emphasis on the translational potential of targeting metabolic pathways influenced by METTL3. The findings from Chen et al. encourage further exploration into therapeutic strategies aimed at disrupting the m^6A modification pathway. Such strategies could potentially enhance the efficacy of existing chemotherapeutic agents and restore sensitivity in previously resistant osteosarcoma cases, highlighting the therapeutic relevance of this study.
Further investigations into the molecular interactions and downstream effects of METTL3 in osteosarcoma will likely yield important insights. Understanding the precise regulatory networks involving m^6A modifications could unveil additional targets for pharmacological intervention. Identifying specific inhibitors of METTL3 or downstream signaling nodes could provide a multi-faceted approach to overcoming chemotherapy resistance and improving patient outcomes.
The utilization of advanced technologies in the study, including CRISPR/Cas9 gene editing and RNA-sequencing, enables a comprehensive analysis of gene expression and regulation. These methodologies provide robust platforms for dissecting the contributions of specific m^6A modifications in real-time. It is crucial for future studies to continue leveraging such technologies to ascertain the full spectrum of genes governed by METTL3 and their roles across various cancer types.
As research in this area grows, the integration of personalized medicine principles will become increasingly critical. Understanding individual genetic backgrounds and tumor characteristics can help tailor treatments that effectively target the specific molecular aberrations present in each patient’s cancer. Such a tailored approach could enhance treatment responses and reduce the incidence of chemoresistance—a critical consideration in the management of osteosarcoma.
In conclusion, the research spearheaded by Chen et al. represents a significant advancement in the understanding of osteosarcoma biology. By elucidating the roles of METTL3, m^6A modification, and calcium signaling in tumor progression, the study presents not only novel insights but also a roadmap for future therapeutic targets. As the scientific community continues to unravel the complexities of cancer epigenetics, the hope is that these discoveries will translate into actionable strategies that can improve the prognosis for patients grappling with the challenges of osteosarcoma.
The impact of such research extends beyond osteosarcoma, as the fundamental mechanisms by which m^6A modifications influence cellular behaviors are pertinent across various cancer types. Future studies should aim to delineate these pathways further, potentially unveiling universal mechanisms that underscore tumor progression and therapeutic resistance, which could inform treatment modalities for a wide array of malignancies.
As this exciting field of research evolves, collaboration between molecular biologists, oncologists, and pharmacologists will be essential in accelerating the transition of these discoveries from the laboratory bench to clinical application. By fostering an interdisciplinary approach, researchers can work towards closing the gap between understanding cancer biology and developing innovative, effective treatments that improve patient outcomes and quality of life in the face of overwhelming odds.
Researchers now face the challenge of integrating these findings into clinical practice, ensuring that the knowledge derived from the laboratory is effectively translated into novel therapies and treatment regimens. The journey from scientific discovery to clinical utility can be arduous, but the advancements highlighted in this study are a testament to the potential for targeted therapies that can change the trajectory of treatment for patients suffering from osteosarcoma and other malignancies.
Furthermore, the implications of targeting the pathways elucidated by Chen et al. resonate within the broader context of cancer treatment. Future studies may well discover additional layers of complexity and opportunities for intervention, fostering new avenues for research that will enhance our understanding of the intricate interplay between genetic modifications and cancer cell survival.
Ultimately, the pursuit of therapeutic breakthroughs in osteosarcoma and beyond hinges on a collaborative spirit among researchers, clinicians, and patients alike. As we strive to navigate the complex landscape of cancer biology, we remain committed to advancing scientific knowledge and translating these insights into meaningful outcomes for those affected by this devastating disease. The research landscape is poised for transformation, and with continued dedication and innovation, we hold the promise of a future where cancer is no longer synonymous with despair, but rather with hope and resilience.
Subject of Research: METTL3-mediated m^6A modification in osteosarcoma
Article Title: METTL3-mediated m^6A modification of CACNA1E promotes osteosarcoma progression and chemoresistance by enhancing WNT7B-mediated Ca^2+ signaling.
Article References:
Chen, C., Xiong, K., Liang, F. et al. METTL3-mediated m6A modification of CACNA1E promotes osteosarcoma progression and chemoresistance by enhancing WNT7B-mediated Ca2+ signaling.
Mol Cancer (2026). https://doi.org/10.1186/s12943-025-02553-x
Image Credits: AI Generated
DOI:
Keywords: m^6A modification, METTL3, osteosarcoma, WNT signaling, chemoresistance.
Tags: CACNA1E gene regulationcalcium signaling in cancerchemoresistance in bone cancerm6A modification in osteosarcomaMETTL3 methyltransferase rolemolecular mechanisms of osteosarcomaoncogenic pathways in cancerosteosarcoma progression mechanismspediatric bone malignancy researchRNA methylation in tumor biologytherapeutic targets for osteosarcomaWNT signaling pathway involvement