Credit: Walsh NM, et al. (2019)
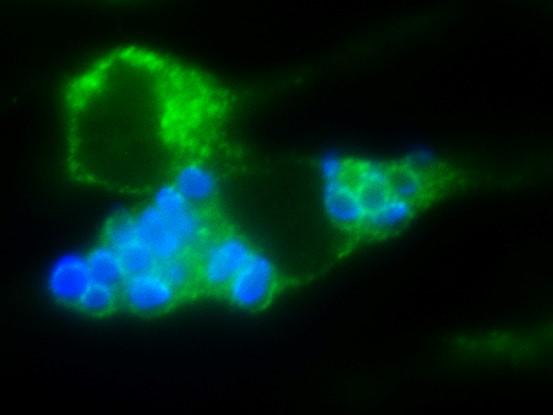
Pathogenic fungal spores capitalize on host immune cells to escape the lung and gain access to the brain to cause fatal disease in mice, according to a study published June 27 in the open-access journal PLOS Pathogens by Christina Hull of the University of Wisconsin-Madison, and colleagues. These insights into the interactions between pathogenic fungal spores and lung immune cells provide new opportunities for understanding spore-mediated fungal diseases.
Little is known about how inhaled spores from human fungal pathogens cause infections and spread to other parts of the body. The most frequent cause of inhaled fatal fungal disease is Cryptococcus, which causes meningitis. To understand how Cryptococcus causes disease, Hull and her colleagues evaluated two types of cells (spores and yeast) in a mouse model of infection. They compared yeast strains that cannot cause disease to the spore offspring they produced during sexual reproduction.
They discovered that parental yeast that are not virulent produced spores that were fully virulent and caused fatal meningitis in 100% of the mice. This difference was associated with movement of spores to the lymph system; mice infected with spores had Cryptococcus in their lung-draining lymph nodes, but mice infected with yeast did not. Furthermore, when they infected mice that lacked immune cells in their lungs, no spores were found in their lymph nodes. This indicates that instead of protecting mice from the spore infection, the immune cells moved spores out of the lung to the lymph system where spores could then spread to the brain. According to the authors, the findings could open new avenues for the development of novel therapeutics that could be effective in the prevention of fatal cryptococcosis and other diseases caused by the spores of invasive human fungal pathogens.
“Inhaling fungal spores causes serious, and even fatal, infections more often than most people realize,” adds Hull. “By understanding how spores move from the lungs to other tissues, we can develop new strategies for preventing spore-mediated fungal diseases and learn how to treat patients more effectively.”
###
Research Article
Funding: This work was supported with NIH grants T32GM007215 (NMW and MRB), R01AI035681 (MW and BK), R01AI093553 (MW and BK), R01AI089370 (CMH) and R01AI137409 (CMH). Further funding was provided to SCO via an HHMI Gilliam Fellowship and to CMH from The Hartwell Foundation Individual Biomedical Research Award, UW SMPH funds, Vilas Life Cycle Award and the Wisconsin Alumni Research Foundation (WARF) funds. Flow data generated at the UWCCC Flow Cytometry Facility was supported by the University of Wisconsin Carbone Cancer Center Support Grant P30 CA014520. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests: The authors have declared that no competing interests exist.
Citation: Walsh NM, Botts MR, McDermott AJ, Ortiz SC, Wüthrich M, Klein B, et al. (2019) Infectious particle identity determines dissemination and disease outcome for the inhaled human fungal pathogen Cryptococcus. PLoS Pathog 15(6): e1007777. https:/
Author Affiliations:
Department of Biomolecular Chemistry, School of Medicine and Public Health, University of Wisconsin-Madison, Madison, Wisconsin, United States of America
Department of Medical Microbiology and Immunology, School of Medicine and Public Health, University of Wisconsin-Madison, Madison, Wisconsin, United States of America
Department of Pediatrics, School of Medicine and Public Health, University of Wisconsin-Madison, Madison, Wisconsin, United States of America
In your coverage please use this URL to provide access to the freely available paper: http://journals.
Media Contact
Christina M. Hull
[email protected]
Original Source
http://journals.
Related Journal Article
http://dx.