Development of 3D electrochemical model to improve battery energy density and efficiency of electric vehicles
Credit: DGIST
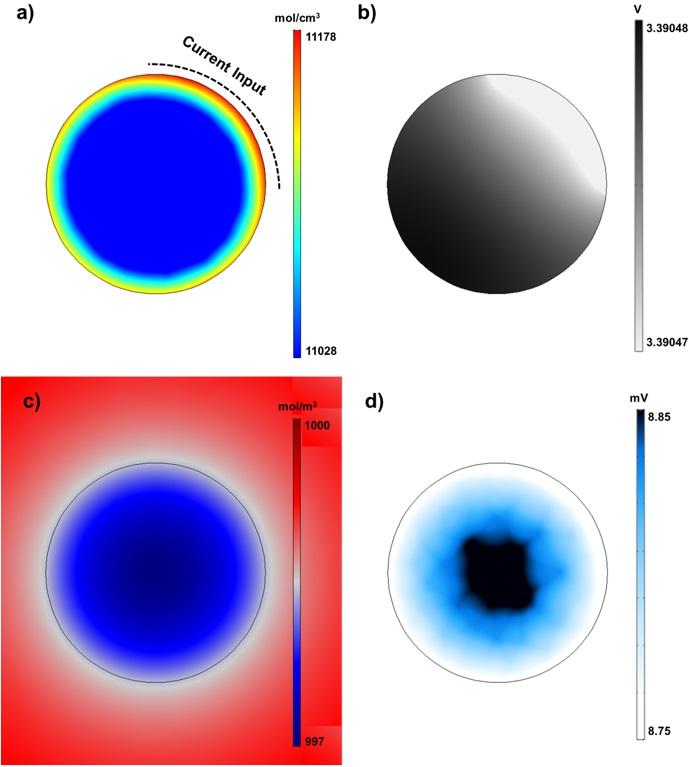
A model that can have a 3D observation of micrometer-sized particles in a cell has been developed. Through the analysis and research of micrometer-sized particles in a cell, this model is expected to enhance energy efficiency of cells.
DGIST announced that Professor Yong Min Lee’s team in the Department of Energy Science & Engineering developed ‘micron1 single particle electrochemical model’ that can estimate the electrochemical properties of a single particle of electrode active materials2 in 3D. The 3D observations of the single particles of electrode active materials, which are difficult to be identified in an experiment, are expected to be applied to research electrochemical phenomena and particle designs that enhance cell efficiency.
Although a secondary cell is commonly used as the power source of electric vehicles, it is still not as efficient as internal combustion engine. Its efficiency can be improved by increasing the energy density of the cells, R&D has not been actively carried out due to the limitations in precise analysis technology.
Professor Lee’s team thought that the energy density of a cell can be enhanced through the design optimization of electrode active materials in a cell. Then, they sought a way to examine the micrometer-sized single particles of electrode active materials and developed electrochemical model that can conduct 3D analysis on the single particles.
Unlike the existing model that focused on cell electrode, the model developed by Professor Yong Min Lee’s team focused on the single particles of active materials that compose electrode. By doing so, the team took another step closer to a research to fundamentally increase cell efficiency through accurate analysis on the properties and characteristics of 3D single particles in a model. Since it can have 3D analysis of particles, the model is especially expected to be applied widely in research to design the single particles of electrode active materials in a cell.
Regarding this research, Professor Yong Min Lee in the Department of Energy Science and Engineering said “Comparing to previous works, our model can look into what happens within a single particle. As a result, it provides an innovative way in designing micrometer-sized particles. Our next goal is to apply this electrochemical model to improve the cell efficiency of electric vehicles.”
###
This research was carried out by Jihun Song (who is currently an integrated M.S.-Ph.D. candidate) and Joonam Park (a Ph.D. candidate) as the co-authors, and it was jointly conducted with Tokyo Metropolitan University (Professor Hirokazu Munakata and Professor Kiyoshi Kanamura), Chungnam National University (Professor Sung-Soo Kim), and Hanbat National University (Professor Myung-Hyun Ryou). The result was published on June 4th in the online version of Nano Energy, an international journal on energy materials.
1 Micron: A length unit defined in 10-6 m and is marked as μm
2 Electrode active material: A material involved in an electrode reaction of a cell
Media Contact
Yong Min Lee
[email protected]
Original Source
https:/
Related Journal Article
http://dx.




