‘Artificial leaf’ concept inspires Rice University research into solar-powered fuel production
Credit: Illustration by Jia Liang/Rice University
HOUSTON – (May 4, 2020) – Rice University researchers have created an efficient, low-cost device that splits water to produce hydrogen fuel.
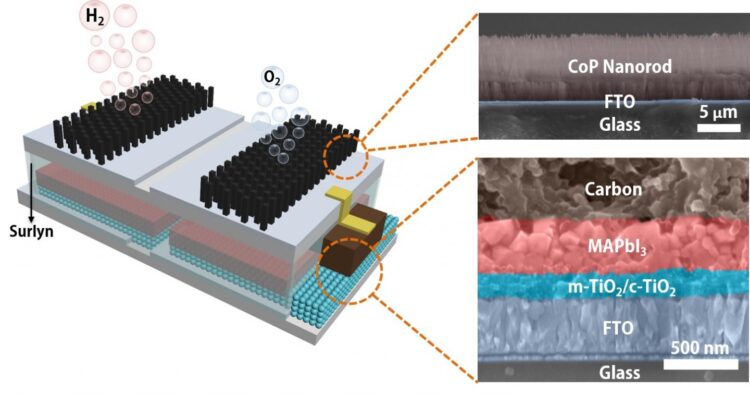
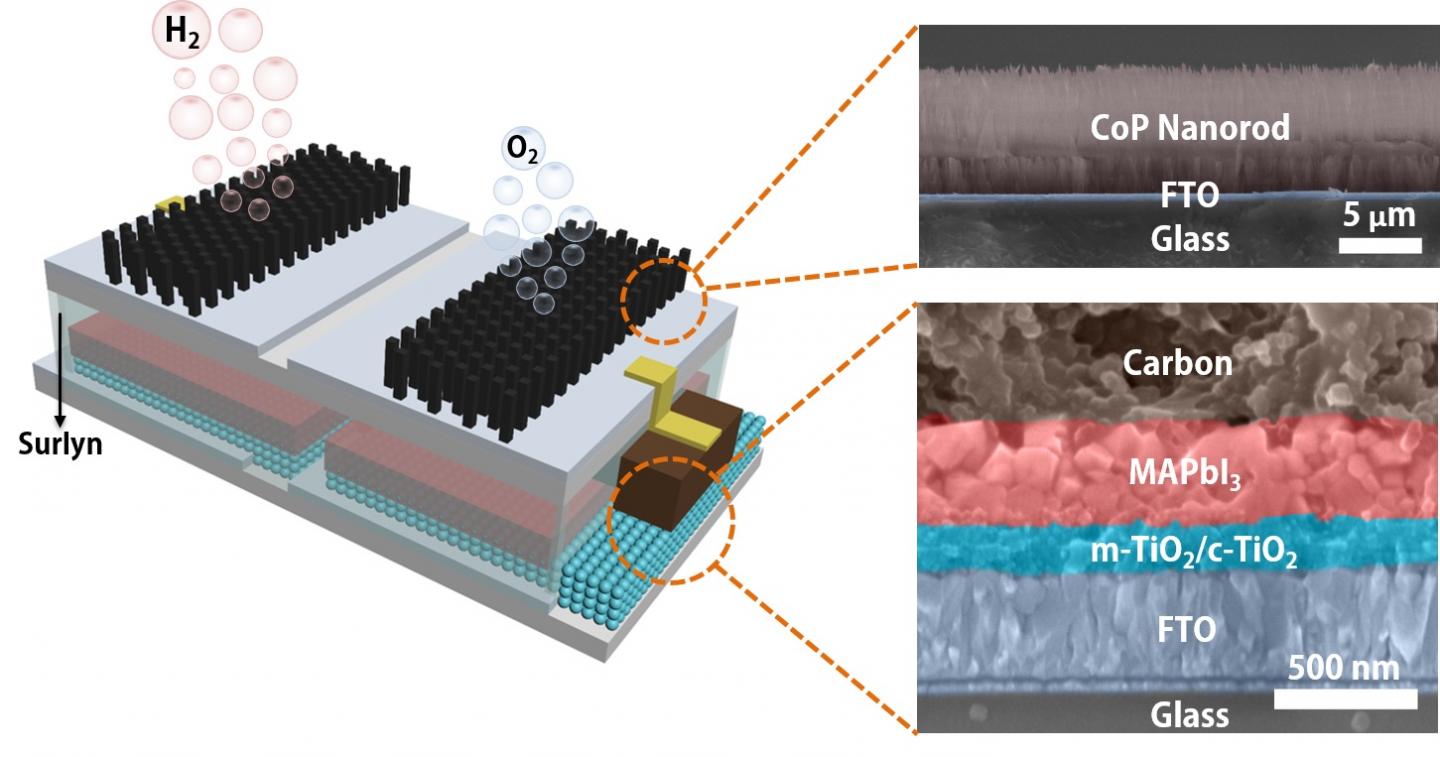
The platform developed by the Brown School of Engineering lab of Rice materials scientist Jun Lou integrates catalytic electrodes and perovskite solar cells that, when triggered by sunlight, produce electricity. The current flows to the catalysts that turn water into hydrogen and oxygen, with a sunlight-to-hydrogen efficiency as high as 6.7%.
This sort of catalysis isn’t new, but the lab packaged a perovskite layer and the electrodes into a single module that, when dropped into water and placed in sunlight, produces hydrogen with no further input.
The platform introduced by Lou, lead author and Rice postdoctoral fellow Jia Liang and their colleagues in the American Chemical Society journal ACS Nano is a self-sustaining producer of fuel that, they say, should be simple to produce in bulk.
“The concept is broadly similar to an artificial leaf,” Lou said. “What we have is an integrated module that turns sunlight into electricity that drives an electrochemical reaction. It utilizes water and sunlight to get chemical fuels.”
Perovskites are crystals with cubelike lattices that are known to harvest light. The most efficient perovskite solar cells produced so far achieve an efficiency above 25%, but the materials are expensive and tend to be stressed by light, humidity and heat.
“Jia has replaced the more expensive components, like platinum, in perovskite solar cells with alternatives like carbon,” Lou said. “That lowers the entry barrier for commercial adoption. Integrated devices like this are promising because they create a system that is sustainable. This does not require any external power to keep the module running.”
Liang said the key component may not be the perovskite but the polymer that encapsulates it, protecting the module and allowing to be immersed for long periods. “Others have developed catalytic systems that connect the solar cell outside the water to immersed electrodes with a wire,” he said. “We simplify the system by encapsulating the perovskite layer with a Surlyn (polymer) film.”
The patterned film allows sunlight to reach the solar cell while protecting it and serves as an insulator between the cells and the electrodes, Liang said.
“With a clever system design, you can potentially make a self-sustaining loop,” Lou said. “Even when there’s no sunlight, you can use stored energy in the form of chemical fuel. You can put the hydrogen and oxygen products in separate tanks and incorporate another module like a fuel cell to turn those fuels back into electricity.”
The researchers said they will continue to improve the encapsulation technique as well as the solar cells themselves to raise the efficiency of the modules.
###
Co-authors of the paper are former Rice visiting students Xiao Han, now an associate professor at Northwestern Polytechnical University in Xi’an, China, and Yunxiu Qiu, now a graduate student at Washington University in St. Louis; Rice graduate students Qiyi Fang and Boyu Zhang; Rice alumnus Weipeng Wang, now an assistant professor at Tsinghua University; Rice postdoctoral researcher Jing Zhang; and Pulickel Ajayan, chair of Rice’s Department of Materials Science and NanoEngineering, the Benjamin M. and Mary Greenwood Anderson Professor in Engineering and a professor of chemistry. Lou is a professor of materials science and nanoengineering and of chemistry.
The research was funded by the Peter M. and Ruth L. Nicholas Postdoctoral Fellowship in Nanotechnology from Rice’s Smalley-Curl Institute, the Welch Foundation, the National Science Foundation-backed Nanosystems Engineering Research Center for Nanotechnology-Enabled Water Treatment, and Fundamental Research Funds for the Central Universities, China.
Read the abstract at https:/
This news release can be found online at https:/
Follow Rice News and Media Relations via Twitter @RiceUNews.
Related materials:
Lou Group: https:/
Department of Materials Science and NanoEngineering: https:/
George R. Brown School of Engineering: https:/
Images for download:
https:/
A schematic and electron microscope cross-section show the structure of an integrated, solar-powered catalyst to split water into hydrogen fuel and oxygen. The module developed at Rice University can be immersed into water directly to produce fuel when exposed to sunlight. (Credit: Illustration by Jia Liang/Rice University)
Located on a 300-acre forested campus in Houston, Rice University is consistently ranked among the nation’s top 20 universities by U.S. News & World Report. Rice has highly respected schools of Architecture, Business, Continuing Studies, Engineering, Humanities, Music, Natural Sciences and Social Sciences and is home to the Baker Institute for Public Policy. With 3,962 undergraduates and 3,027 graduate students, Rice’s undergraduate student-to-faculty ratio is just under 6-to-1. Its residential college system builds close-knit communities and lifelong friendships, just one reason why Rice is ranked No. 1 for lots of race/class interaction and No. 4 for quality of life by the Princeton Review. Rice is also rated as a best value among private universities by Kiplinger’s Personal Finance.
Jeff Falk
713-348-6775
[email protected]
Mike Williams
713-348-6728
[email protected]
Media Contact
Jeff Falk
[email protected]
Original Source
https:/
Related Journal Article
http://dx.