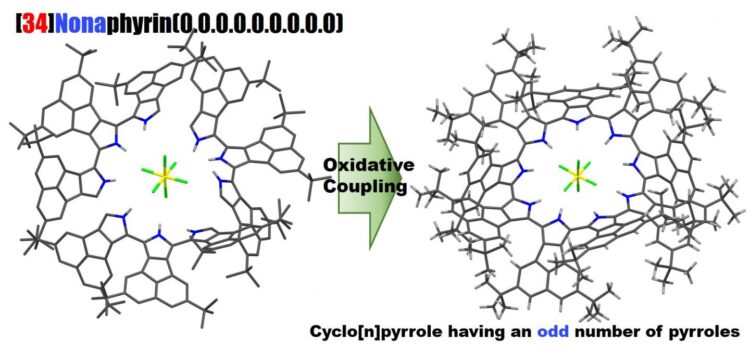
Selective synthesis of ring-expanded porphyrinoids comprising nine pyrroles
Profs. Okujima and Uno at Ehime University, in collaboration with Prof. Kobayashi at Shinshu University, reported the selective synthesis, the molecular structure, optical properties and electronic structure of cyclo[9]pyrrole, a ring-expanded porphyrin consisting of directly connected pyrrole rings.
We successfully synthesized a good yield of cyclo[9]pyrroles via the oxidative coupling of terpyrrole. A relatively distorted structure with a C2-like symmetry was clarified by NMR and X-ray diffraction analyses. Intense absorption was observed at ca. 1,740 nm. We analyzed the optical and electronic structures using magnetic circular dichroism spectroscopy and time-dependent density functional theory calculations. Comparison of cyclo[8], [9], and [10]pyrroles showed the electronic structures don’t significantly depend on the number of pyrroles.
###
Our findings were published on April 15, 2021 in Organic Letters.
Media Contact
Public Relations Division
[email protected]
Original Source
https:/
Related Journal Article
http://dx.