Credit: Lei Dong, Nanjing University
Scientists from Nanjing University and the University of Macau have devised a new approach to extend the survival of transplanted probiotics in vivo, enhancing the efficacy of cancer chemo-/immunotherapies in mice. The paper entitled “Smectite promotes probiotic biofilm formation in the gut for cancer immunotherapy” appears online today in Cell Reports.
The gut contains trillions of symbiotic bacteria. Disturbing the balance of intestinal flora may increase the occurrence of major diseases, including cancers. The gut microbiome plays an essential role in regulating the host immunity, which has inspired strategies to modulate intestinal microorganisms for augmenting cancer chemo-/immunotherapy. Studies have shown that probiotics such as Lactobacillus and Bifidobacterium in intestinal flora can activate host immunity and sensitise tumour immunotherapy. Nevertheless, the two major approaches to change the gut microbiome’s composition have met with substantial challenges: oral administration of probiotics is often inefficient, and faecal microbiota transplantation (FMT) risks pathogen infection. Therefore, scientists are looking for new and safe strategies to enrich probiotics in the host intestine.
The study found that Smectite, a drug commonly used for treating gastrointestinal diseases such as diarrhoea, could selectively promote the formation of lactic acid bacteria (LAB) biofilms, which foster LAB colonisation in the intestinal tract. The increase of probiotics in the intestinal tract induces anti-tumour immune responses, thereby inhibiting tumour growth and improving immunotherapy efficacy.
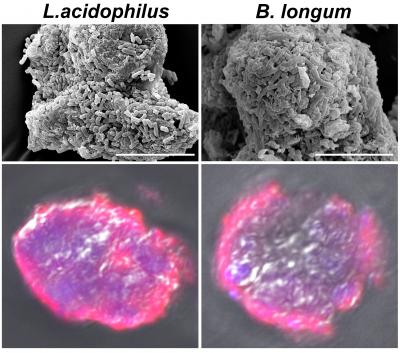
The researchers found that Smectite treatment increased Lactobacillus and Bifidobacterium in mice guts. Meanwhile, electron microscopy showed that only Lactobacillus and Bifidobacterium could form biofilms on the surface of Smectite, thanks to the latter’s ion-exchanging microstructure. The researchers then demonstrated that oral Smectite exerted anti-tumour effects by altering the intestinal flora of mice. Moreover, considering the low number of beneficial bacteria in the gut, the researchers constructed smectite-lactobacillus-biofilm microspheres (SLB) in vitro. They fed the melanoma model mice with these microspheres, and the SLB showed a more significant anti-tumour effect than using Smectite or Lactobacillus alone.
The team further discovered a possible mechanism that SLB could stimulate dendritic cells’ maturation (DCs) through the toll-like receptor 2 (TLR2) signalling. Meanwhile, the proliferation of CD8+T cells was stimulated, and the expression of IFN-γ and IL12 increased. Intriguingly, SLB drove the mature DCs from the colon to the tumour tissue and play the anti-tumour effect. Finally, the researchers further used the SLB in combination with chemotherapy and immunotherapy. In a mouse model, the combination of SLB with doxorubicin or anti- PD-1 antibodies showed better anti-tumour efficacy compared with using SLB, doxorubicin, or anti-PD-1 antibody alone.
This study provides an effective and safe strategy to promote probiotic colonisation and expansion in gut microbiota, via the specific support of Smectite for probiotic biofilm formation. The smectite-aided probiotic treatment was effective in activating the anti-tumour immune responses of DCs and cytotoxic T cells. According to the lead author, Prof Lei Dong, because Smectite has been clinical used for half a century in many countries – albeit, for another application – its safety data may be readily found to accelerate its translation for immunotherapy purpose. This gut microbiome-based, DC-activating therapeutic approach may have immediate translational potential for cancer immunotherapy.
###
Media Contact
Lei Dong
[email protected]
Related Journal Article
http://dx.