One in a thousand children in the United States is deaf, and one in three adults will experience significant hearing loss after the age of 65. Whether the result of genetic or environmental factors, hearing loss costs billions of dollars in healthcare expenses every year, making the search for a cure critical.
Now a team of researchers led by Karen B. Avraham of the Department of Human Molecular Genetics and Biochemistry at Tel Aviv University’s Sackler Faculty of Medicine and Yehoash Raphael of the Department of Otolaryngology–Head and Neck Surgery at University of Michigan’s Kresge Hearing Research Institute have discovered that using DNA as a drug — commonly called gene therapy — in laboratory mice may protect the inner ear nerve cells of humans suffering from certain types of progressive hearing loss.
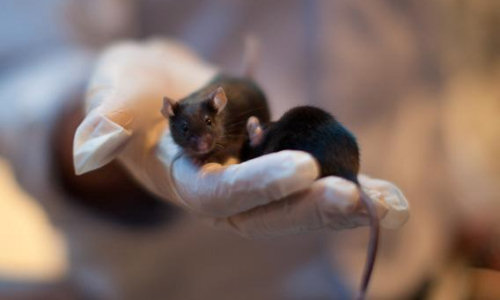
In the study recently published in the journal Hearing Research, doctoral student Shaked Shivatzki created a mouse population possessing the gene that produces the most prevalent form of hearing loss in humans: the mutated connexin 26 gene. Some 30 percent of American children born deaf have this form of the gene. Because of its prevalence and the inexpensive tests available to identify it, there is a great desire to find a cure or therapy to treat it.
“Regenerating” neurons
Prof. Avraham’s team set out to prove that gene therapy could be used to preserve the inner ear nerve cells of the mice. Mice with the mutated connexin 26 gene exhibit deterioration of the nerve cells that send a sound signal to the brain. The researchers found that a protein growth factor used to protect and maintain neurons, otherwise known as brain-derived neurotrophic factor (BDNF), could be used to block this degeneration. They then engineered a virus that could be tolerated by the body without causing disease, and inserted the growth factor into the virus. Finally, they surgically injected the virus into the ears of the mice. This factor was able to “rescue” the neurons in the inner ear by blocking their degeneration.
“A wide spectrum of people are affected by hearing loss, and the way each person deals with it is highly variable,” said Prof. Avraham. “That said, there is an almost unanimous interest in finding the genes responsible for hearing loss. We tried to figure out why the mouse was losing cells that enable it to hear. Why did it lose its hearing? The collaborative work allowed us to provide gene therapy to reverse the loss of nerve cells in the ears of these deaf mice.”
Although this approach is short of improving hearing in these mice, it has important implications for the enhancement of sound perception with a cochlear implant, used by many people whose connexin 26 mutation has led to impaired hearing.
Embryonic hearing?
Inner ear nerve cells facilitate the optimal functioning of cochlear implants. Prof. Avraham’s research suggests a possible new strategy for improving implant function, particularly in people whose hearing loss gets progressively worse with time, such as those with profound hearing loss as well as those with the connexin gene mutation. Combining gene therapy with the implant could help to protect vital nerve cells, thus preserving and improving the performance of the implant.
More research remains. “Safety is the main question. And what about timing? Although over 80 percent of human and mouse genes are similar, which makes mice the perfect lab model for human hearing, there’s still a big difference. Humans start hearing as embryos, but mice don’t start to hear until two weeks after birth. So we wondered, do we need to start the corrective process in utero, in infants, or later in life?” said Prof. Avraham.
“Practically speaking, we are a long way off from treating hearing loss during embryogenesis. But we proved what we set out to do: that we can help preserve nerve cells in the inner ears of the mouse,” Prof. Avraham continued. “This already looks very promising.”
The research team is currently working on finding better “vehicles” for the corrected gene, such as finding more suitable viruses to transport the injected gene to the appropriate place in the inner ear.
Story Source:
The above story is based on materials provided by Tel Aviv University.