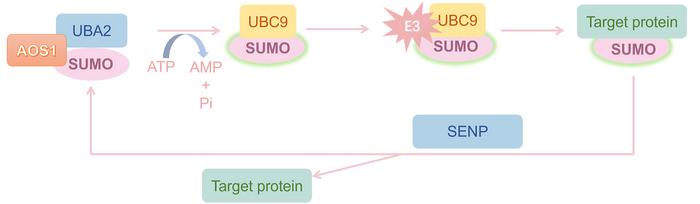
The nuclear pore complex protein RANBP2 has recently gained significant attention within the realm of cancer biology due to its critical function as a SUMO E3 ligase, orchestrating the post-translational modification known as SUMOylation. This biochemical process involves the covalent attachment of Small Ubiquitin-like Modifier (SUMO) proteins to target substrates, profoundly impacting cellular processes such as protein stability, localization, and activity. RANBP2’s role in modulating the cell cycle through SUMOylation cements its position as a vital regulatory node, with emerging research cataloging its extensive involvement in the pathogenesis of diverse solid tumors.
SUMOylation, distinct yet mechanistically akin to ubiquitination, serves as a molecular switch controlling a plethora of oncogenes and tumor suppressors. The strategic placement of RANBP2 within the nuclear pore complex is pivotal, as it not only facilitates nucleocytoplasmic trafficking but also governs the fidelity of mitotic progression. This dual functionality underscores RANBP2’s capacity to influence cellular homeostasis and, when dysregulated, contribute to oncogenic transformation. Recent insights delineate RANBP2’s multifaceted roles in cancers such as hepatocellular carcinoma, gastric and breast cancers, among others, highlighting its potential as a molecular lynchpin in tumor biology.
In hepatocellular carcinoma (HCC), RANBP2 exerts a profound effect by SUMOylating LASP1, a protein associated with cytoskeletal dynamics and cellular motility. This modification upregulates HER2 expression, fostering an environment conducive to unchecked proliferation and tumor expansion. Beyond this, RANBP2 modulates the transcription factor NR5A2, leading to altered alpha-fetoprotein levels, a clinically relevant biomarker in HCC diagnosis. Additionally, RANBP2’s SUMOylation of IL-33 has been implicated in immune evasion strategies of HCC cells, presenting a sophisticated interplay between tumor progression and immune escape mechanisms.
Cholangiocarcinoma, a notoriously aggressive malignancy of the biliary tract, also manifests aberrant RANBP2 activity. Here, the SUMOylation of p27kip1 induces its translocation to the nucleus, disrupting cell cycle checkpoints and encouraging tumor cell proliferation. This nuclear relocalization of p27kip1, ordinarily a cyclin-dependent kinase inhibitor, reveals how post-translational modifications mediated by RANBP2 can invert traditional tumor suppressive functions, thereby facilitating oncogenesis.
The oncogenic influence of RANBP2 extends to gastric cancer, where it interacts with the death domain-associated protein DAXX. This interaction promotes DAXX nuclear localization, which has been correlated with poor prognosis and aggressive tumor phenotypes. DAXX’s nuclear functions, including transcriptional regulation and chromatin remodeling, when exacerbated by enhanced SUMOylation signatures, contribute to the epigenetic dysregulation observed in gastric carcinoma.
In breast cancer, the SUMOylation landscape shaped by RANBP2 proves equally consequential. Modification of β-arrestin 2 disrupts the critical MDM2-p53 signaling axis, a pathway central to genomic stability and apoptosis. Through this disruption, p53 activity is paradoxically enhanced, leading to tumor suppression. This nuanced role of RANBP2 spotlights its capacity to wield dualistic effects, potentially constraining tumor growth depending on cellular context and substrate specificity.
RANBP2 also orchestrates tumor progression in cervical cancer by enhancing the transcriptional activity of TCF4 through SUMOylation. This activation ultimately fuels the Wnt/β-catenin signaling pathway, renowned for driving cell proliferation, invasion, and metastasis in many cancers. The biochemical modifications introduced by RANBP2 reinforce this oncogenic signaling cascade, cementing its role in disease advancement.
Similarly, in prostate cancer, RANBP2 modulates p53 SUMOylation status, intricately influencing androgen receptor-mediated pathways. Given the androgen receptor’s pivotal role in prostate cancer biology, RANBP2’s regulatory function here affects cancer cell proliferation and survival, suggesting that disrupting this axis may offer therapeutic benefit.
The oncogenic relevance of RANBP2 is not confined to these malignancies. In glioblastoma, a deadly brain tumor with dismal prognosis, RANBP2-driven SUMOylation events have been linked to DNA repair and chromatin reorganization mechanisms critical for tumor survival. The protein’s influence on genomic stability pathways indicates potential vulnerability points for targeted intervention.
Further, emerging evidence points to RANBP2’s involvement in oral and colorectal cancers. In colorectal cancer, its depletion destabilizes the mitotic spindle apparatus, provoking apoptosis and hampering tumor growth. This suggests that modulation of RANBP2 activity may disrupt cell division fidelity, a hallmark of cancer cells. In lung cancer, RANBP2’s interaction with DNA Topoisomerase II, an enzyme vital for DNA replication and chromosomal segregation, hints at its broader role in maintaining genetic integrity during rapid tumor cell proliferation.
The cumulative understanding of RANBP2’s diverse interactions and regulatory functions underscores its attractiveness as a therapeutic target. However, the intricate network of molecular mechanisms modulated by this SUMO E3 ligase demands comprehensive research to deconvolute its context-dependent effects and to develop selective inhibitors that exploit its oncogenic vulnerabilities without disrupting essential cellular processes.
Targeting post-translational modifiers like RANBP2 epitomizes a frontier in cancer therapeutics, offering avenues for precision medicine aimed at debilitating core molecular machinery of tumor cells. As ongoing studies unravel the complexity of SUMOylation landscapes in different tumor microenvironments, RANBP2 stands out as a promising candidate for novel drug development strategies capable of impeding cancer progression and improving clinical outcomes.
With the accelerated pace of discovery in molecular oncology and functional proteomics, elucidating the full repertoire of RANBP2-modified substrates and their downstream pathways is imperative. This knowledge will pave the way for the rational design of SUMOylation modulators and combinatorial approaches that can effectively shut down cancer-promoting circuits orchestrated by RANBP2.
In conclusion, RANBP2’s role as a central SUMO E3 ligase within the nuclear pore complex places it at the nexus of multiple tumorigenic processes spanning cell cycle control, protein localization, and gene expression regulation. Its multifarious engagement across a spectrum of solid malignancies highlights its potential both as a biomarker and as a therapeutic target. The translation of these molecular insights into clinical applications may revolutionize treatment paradigms for several aggressive cancers in the near future.
Subject of Research: Nuclear pore complex protein RANBP2 and its role in SUMOylation in solid malignancies.
Article Title: Nuclear pore complex protein RANBP2 and related SUMOylation in solid malignancies.
News Publication Date: Not specified; article volume indicates 2025.
Web References: http://dx.doi.org/10.1016/j.gendis.2024.101407
References:
Xinning Yu, Huatao Wu, Zheng Wu, Yangzheng Lan, Wenjia Chen, Bingxuan Wu, Yu Deng, Jing Liu, Nuclear pore complex protein RANBP2 and related SUMOylation in solid malignancies, Genes & Diseases, Volume 12, Issue 4, 2025, 101407.
Image Credits: Genes & Diseases
Keywords: RANBP2, SUMOylation, nuclear pore complex, solid malignancies, hepatocellular carcinoma, gastric cancer, breast cancer, cervical cancer, prostate cancer, glioblastoma, colorectal cancer, lung cancer, post-translational modification, cancer therapeutics.
Tags: dual functionality in mitotic progressiondysregulation of cellular homeostasishepatocellular carcinoma researchmolecular targets in cancer treatmentnuclear pore complex proteinsoncogenic transformation mechanismsprotein stability and localizationRANBP2 role in cancer therapyroles in breast and gastric cancersSUMO E3 ligase functionSUMOylation in solid tumorstumor biology and pathogenesis