New method is 1000 times faster and could be used to illuminate reaction mechanisms behind artificial photosynthesis
Credit: (TOC graphic from the ACS Catalyst journal paper).
Currently, the majority of energy consumed by the world’s population is derived from oil and other non-renewable resources which are in danger of running out in the near future. Consequently the development of artificial photosynthesis methods using photocatalysts to produce chemical energy (hydrogen fuel) from sunlight and water has received much attention and various research projects are being conducted in this area.
During artificial photosynthesis, oxygen (O2) is produced by the photocatalyst via the water splitting reaction. Working with researchers from Kanazawa University, Shinshu University and The University of Tokyo, Professor ONISHI Hiroshi et al. of Kobe University’s Graduate School of Science developed a measurement evaluation method which is able to detect O2 1000 times faster than conventional methods. It is hoped that the method developed through this research can be utilized to improve our understanding of the reaction mechanisms behind artificial photosynthesis and contribute towards developing photocatalysts that could be implemented in the real world.
The importance of making these research results public as soon as possible has been recognized; the paper published in the American Chemistry Society’s journal ACS Catalysis was given an advanced online release on October 29, 2020.
Research Background
Artificial photosynthesis, which can be utilized to produce chemical energy (hydrogen fuel) from sunlight and water has received much attention for its potential to provide an energy source that does not emit CO2. Photocatalysts are the key component of artificial photosynthesis. The first photocatalyst material was discovered and developed by Japanese researchers in the 1970s, and scientists around the world have continuously strived to improve their efficiency over the last 50 years.
The current research study used a strontium titanate (SrTiO3) photocatalyst, which was originally developed by Special Contract Professor DOMEN Kazunari et al. of Shinshu University (a contributing researcher to this study). As a result of various improvements made by Shinshu’s Associate Professor HISATOMI Takashi et al. (also a contributing researcher), this photocatalytic material achieved the highest reaction yield (i.e. the efficiency of hydrogen conversion from water via illumination by ultraviolet light) in the world. The final remaining issue is improving the efficiency of hydrogen generation from water and sunlight, instead of artificial ultraviolet light. Overcoming this issue would mean the birth of CO2-free hydrogen fuel producing technology that can be utilized by society.
However, one factor that hinders efforts to improve conversion efficiency is the low rate of oxygen produced from the water when hydrogen is also being produced. In order to generate hydrogen (H2) from water (H2O) via artificial photosynthesis, the following chemical reaction has to take place: 2H2O -> 2H2 + O2. Even though the goal is to produce hydrogen (that can be utilized as a fuel by society) and not oxygen, the principles of chemistry require oxygen to be produced from the water at the same time in order for hydrogen to be produced.
Furthermore, the process of generating oxygen is more complicated than the process of generating hydrogen, which consequently makes it difficult to improve the efficiency of the reaction (The oxygen atoms taken from 2 H2O particles must adhere to one another.). This is a bottleneck that limits the efficient conversion of hydrogen from water using sunlight.
A solution would be to improve the efficiency of oxygen conversion from water, however this is no simple matter. It is not well understood how oxygen is generated from water (i.e. the mechanism behind the reaction), therefore trying to improve this reaction is akin to working in the dark. In order to shed light on the situation, this research aimed to develop a high speed detection method to observe the oxygen generated by artificial photosynthesis to reveal the mechanism behind the water to oxygen reaction.
Research Methodology
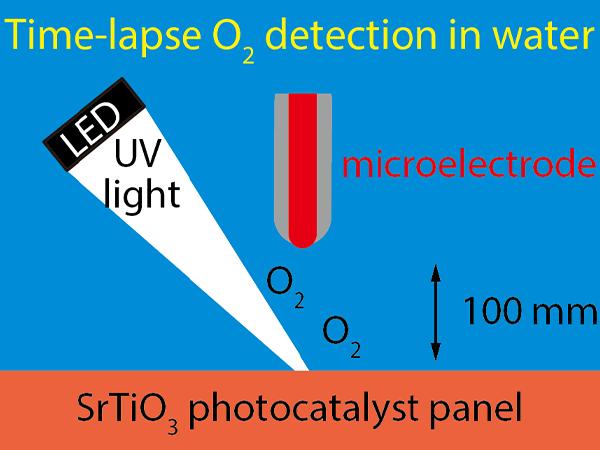
This research study utilized a method of underwater chemical analysis using microelectrode developed by Kanazawa University’s Professor TAKAHASHI Yasufumi et al. (contributing researcher) as the underlying technology. The oxygen generated from the artificial photosynthesis photocatalyst was detected as it merged back into the water. As shown in Figure 1, the strontium titanite photocatalyst panel was submerged in water. The microelectrode, which consisted of a 20 micrometer platinum wire (about ¼ of a human hair) with its sides coated in glass, was lowered into the water 100 micrometers away from the surface of the photocatalyst panel.
When the photocatalyst panel was illuminated by ultraviolet light (with a wavelength of 280nm) from a light-emitting diode, oxygen (O2) and hydrogen (H2) were dissociated from the water where it made contact with the panel. These oxygen and hydrogen molecules were subsequently released into the water. The released oxygen was scattered throughout the water and reached the microelectrode. The oxygen that reached the microelectrode received 4 electrons (e–) from the electrode resulting in the following transformation: O2 + 2H2O + 4e– -> 4OH–.
The number of electrons received from the electrode by the oxygen can be determined by measuring the electric current that passes through the electrode. Measuring the electric current that passed through the electrode every 0.1 seconds enabled the researchers to calculate the amount of oxygen that reached the electrode every 0.1 seconds. Gas-chromatographic detection, the analytic apparatus used for oxygen detection up until now, can only measure the amount of oxygen every 3 minutes. This study succeeded in developing a detection method that is 1000 times faster.
Calculating the time required for the oxygen to travel the 100 micrometer distance through the water from the photocatalyst panel to the electrode is not difficult. This can be achieved by conducting numerical simulations on a desktop computer, based on Fick’s laws of diffusion. Comparing the measurement results obtained from the microelectrode with those of the simulation revealed that there was a 1 to 2 second delay between the photocatalyst panel being illuminated by UV light and the oxygen being released into the water. This delay is a new phenomenon that couldn’t be observed via gas-chromatographic detection.
It is believed that this delay is a necessary preparatory stage for the illuminated photocatalyst to commence water-splitting. Future research will seek to verify this hypothesis, in addition to investigating what the photocatalyst is doing during the preparatory stage. Nevertheless, it is expected that the oxygen detection method developed in this study, which is 1000 times faster than previous detection methods, will lead to new developments in artificial photosynthesis.
Researcher Comment (Professor Onishi Hiroshi, Graduate School of Science, Kobe University)
I am a physical chemistry specialist, and the idea to detect the oxygen generated via artificial photosynthesis using a microelectrode came to me in 2015. At Kobe University, we set up the measuring apparatus developed by Professor Takahashi et al., who are experts in chemical analysis using microelectrodes, and began to apply it to photocatalysts.
By improving the apparatus and accumulating know-how regarding its operation, we verified that this method is able to measure the oxygen generated from the photocatalyst panel provided by Professor Domen and Associate Professor Hisatomi et al., who are authorities on photocatalyst research.
In addition, 3 graduate students at Kobe University’s Graduate School of Science were at the forefront of this research for the 5 year period spanning from the development of the computer program for the numerical simulation up until the discovery of the ‘oxygen release delay’.
The three teams brought the distinct features of their respective fields of physical chemistry, analytical chemistry and catalyst chemistry to the development of this research. Through this collaboration, we succeeded in contributing a new perspective to the science of artificial photosynthesis.
###
Research Funding
This study was supported by funding from the Japan Society for the Promotion of Science (JSPS KAKENHI, grant numbers JP16H02250 and JP19H00915) and the Japan Science and Technology Agency’s PRESTO program (JPMJPR18T8).
Journal Information
Title:
“Transient Kinetics of O2 Evolution in Photocatalytic Water-Splitting Reaction”
DOI: 10.1021/acscatal.0c04115
Authors:
Takumu Kosaka1, Yuya Teduka1, Takuya Ogura1, Yuanshu Zhou2, Takashi Hisatomi3, Hiroshi Nishiyama4, Kazunari Domen3, 4, Yasufumi Takahashi2, 5, Hiroshi Onishi1, *
1 Graduate School of Science, Kobe University.
2 Nano Life Science Institute, Kanazawa University.
3 Interdisciplinary Cluster for Cutting Edge Research, Shinshu University.
4 Office of University Professors, The University of Tokyo.
5 Precursory Research for Embryonic Science and Technology, Japan Science and Technology Agency (JST)
* Corresponding author.
Journal:
ACS Catalysis
Media Contact
Verity Townsend
[email protected]
Original Source
https:/
Related Journal Article
http://dx.




