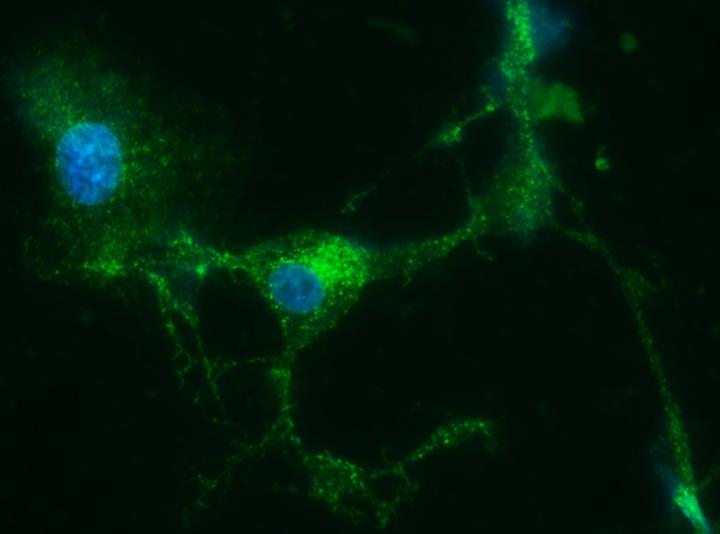
Credit: Choi, et al. (CC BY 4.0, 2018)
Infections by the mosquito-borne Mayaro virus (MAYV) have largely been restricted to people who ventured into the heavily forested areas of Trinidad and Tobago and neighboring regions of South America. However, recent laboratory studies showing additional mosquito vectors can transmit MAYV as well as a reported case of MAYV infection in Haiti in 2015, reveal the potential for the virus to expand its range further into tropical regions of the Caribbean and Central and South America. Now, researchers reporting in PLOS Neglected Tropical Diseases have developed a novel vaccine capable of inducing protective immunity against MAYV.
MAYV, which causes a fever, rash, headache, nausea and vomiting, was first identified in Trinidad in 1954. Some people who contract MAYV develop recurrent joint pain that can last for months after the initial infection. Over the years since its discovery, only sporadic cases have been reported, mostly in tropical areas of South America, but recent surveys have suggested it may be circulating in Central America and a case was confirmed in Haiti in 2015.
MAYV infection causes fever, rash, headache, nausea, and vomiting In the new study, Kar Muthumani, director of the Laboratory of Emerging Infectious Diseases at The Wistar Institute, and colleagues used synthetic DNA technologies to design a DNA vaccine that encodes the sequence for the MAYV envelope (E) glycoproteins. The researchers optimized a consensus sequence representative of multiple MAYV strains (scMAYV-E) to account for the natural genetic variability in the main antigen on the surface of the virus.
The scMAYV-E DNA vaccine was administered by intramuscular injection followed by pulses of electricity designed to make cells more permeable to foreign DNA and enhance vaccine uptake, which allows for dose-sparing.
Immunization with scMAYV-E induced robust MAYV-specific immune responses in mice, including both MAYV infection-neutralizing antibodies as well as cellular responses to multiple regions of MAYV-E. Importantly, the scMAYV-E vaccine provided complete protection from death and clinical signs of infection in a MAYV-challenge mouse model.
“The robust immunogenicity of the scMAYV-E vaccine demonstrated here supports the need for further testing of this vaccine as a viable means to halt the spread of this virus and protect individuals at risk from MAYV disease,” the researchers say. “DNA vaccines have a remarkable safety record in numerous clinical trials, can be designed and manufactured readily, and distributed cost-effectively, making them an important tool for combating emerging infectious diseases like MAYV especially in resource-poor settings, where they often arise.”
###
In your coverage please use this URL to provide access to the freely available paper: http://journals.
Citation: Choi H, Kudchodkar SB, Reuschel EL, Asija K, Borole P, et al. (2019) Protective immunity by an engineered DNA Vaccine for Mayaro Virus. PLOS Neglected Tropical Diseases 13(2): e0007042. https:/
Funding: Kar Muthumani and David B. Weiner note funding by Inovio Pharmaceuticals, PA, USA. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing Interests: Charles Reed, Stephanie Ramos, Laurent Humeau, and J. Joseph Kim are employees of Inovio Pharmaceuticals and as such receive salary and benefits, including ownership of stock and stock options. Kar Muthumani (KM) discloses grant funding, industry collaborations, speaking honoraria, and fees for consulting. He has received consulting fees from Inovio Pharmaceuticals related to DNA vaccine development. He has a patent application for DNA vaccine development and delivery of DNA encoded monoclonal antibodies pending to Inovio Pharmaceuticals. Remuneration includes direct payments. David B. Weiner (DBW) is the W.W. Smith Charitable Trust Professor in Cancer Research at The Wistar Institute. DBW discloses grant funding, SAB and Board service, industry collaborations, speaking honoraria, and fees for consulting. His service includes serving on scientific review committees and advisory boards. Remuneration includes direct payments and/or stock or stock options. He notes potential conflicts associated with this work with Pfizer, Bristol Myers Squibb, Inovio Pharmaceuticals, Merck, VGXI, Geneos, AstraZeneca and potentially others. Licensing of technology from this laboratory has created over 150 jobs in the biotech/pharma industry. The other authors declare no competing financial interests.
Media Contact
Kar Muthumani
[email protected]
Related Journal Article
http://dx.