Although there are medications that can compensate for the loss of female sex hormone production, the drugs are often not recommended for long-term use due to the increased risk of heart disease and breast cancer.
“Our goal is to develop a tissue- or cell-based hormone therapy – essentially an artificial ovary- to deliver sex hormones in a more natural manner than drugs,” said Emmanuel C. Opara, Ph.D., professor of regenerative medicine and senior author. “A bioartificial ovary has the potential to secrete hormones in a natural way based on the body’s needs, rather than the patient taking a specific dose of drugs each day.”
Ovaries are the female reproductive organs that produce eggs that are fertilized for pregnancy as well as secrete hormones important to bone and cardiovascular health. The loss of ovarian function can be due to surgical removal, chemotherapy and radiation treatments for certain types of cancer, and menopause. The effects of hormone loss can range from hot flashes and vaginal dryness to infertility and increased risk of osteoporosis and heart disease.
“This research project is interesting because it offers hope to replace natural ovarian hormones in women with premature ovarian failure or in women going through menopause,” Tamer Yalcinkaya, M.D., associate professor and section head of reproductive medicine at Wake Forest Baptist. “The graft format would bring certain advantages: it would eliminate pharmacokinetic variations of hormones when administered as drugs and would also allow body’s feedback mechanisms to control the release of ovarian hormones.”
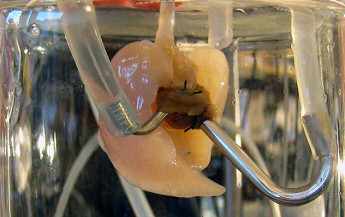
The project to engineer a bioartificial ovary involves encapsulating ovarian cells inside a thin membrane that allows oxygen and nutrients to enter the capsule, but would prevent the patient from rejecting the cells. With this scenario, functional ovarian tissue from donors could be used to engineer bioartificial ovaries for women with non-functioning ovaries.
The Wake Forest Baptist team isolated the two types of endocrine cells found in ovaries (theca and granulosa) from 21-day-old rats. The cells were encapsulated inside materials that are compatible with the body. The scientists evaluated three different ways of arranging the cells inside the capsules.
The function of the capsules was then evaluated in the lab by exposing them to follicle-stimulating hormone and luteinizing hormone, two hormones that stimulate ovaries to produce sex hormones. The arrangement of cells that most closely mimicked the natural ovary (layers of cells in a 3-D shape) secreted levels of estrogen that were 10 times higher than other cell arrangements.
The capsules also secreted progesterone as well as inhibin and activin, two hormones that interact with the pituitary and hypothalamus and are important to the body’s natural system to regulate the production of female sex hormones.
“Cells in the multilayer capsules were observed to function in similar fashion to the native ovaries,” said Opara. “The secretion of inhibin and activin secretion suggests that these structures could potentially function as an artificial ovary by synchronizing with the body’s innate control system.”
Opara said the next step in the research, already underway, is to evaluate the function of the ovarian structures in animals.
Opara’s co-researchers were Sivanandane Sittadjody, Ph.D, Sunyoung Joo, M.D., Ph.D., James J. Yoo, M.D., Ph.D., and Anthony Atala, M.D., all from Wake Forest Baptist, and Justin M. Saul, Ph.D., a former Wake Forest Baptist researcher now at Miami University.
Story Source:
The above story is reprinted from materials provided by Wake Forest Baptist Medical Center