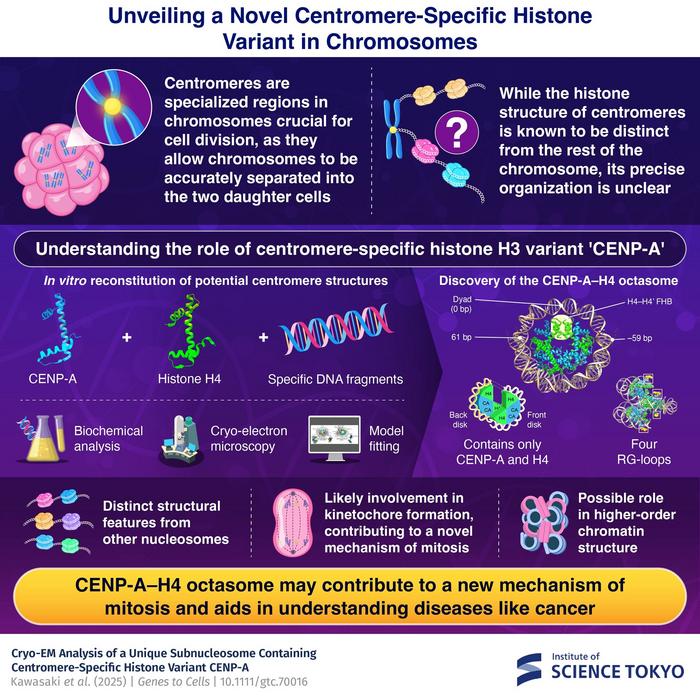
In a groundbreaking study that redefines our understanding of chromosome architecture, researchers from the Institute of Science Tokyo have unveiled an unprecedented structure at the core of chromosome organization known as the centromere. This specialized region, essential for correct segregation of chromosomes during mitosis, has long eluded detailed molecular characterization due to its complex and unique composition. The team, led by Associate Professor Kayo Nozawa, reveals the existence of a novel nucleosome-like particle dubbed the “CENP-A–H4 octasome” — a molecular assembly that radically differs from conventional nucleosomes and promises to illuminate the intricacies of centromere function and chromatin biology.
Centromeres play a pivotal role in cell division by serving as assembly platforms for kinetochores, intricate protein machineries that anchor chromosomes to the spindle fibers, ensuring accurate distribution of genetic material to daughter cells. While it is established that canonical nucleosomes, the fundamental units of chromatin, comprise histone octamers — two each of H2A, H2B, H3, and H4 — wrapped by approximately 145 base pairs of DNA, centromeric chromatin instead contains a distinct histone H3 variant named CENP-A. The precise structural organization of these centromeric nucleosomes and how they facilitate kinetochore assembly have been subjects of intense scientific debate.
Diverging from the canonical nucleosome blueprint, the CENP-A–H4 octasome lacks two of the histone components traditionally present in nucleosomes, specifically H2A and H2B, which are notably underrepresented in yeast centromeres. By reconstituting nucleosome-like particles exclusively with CENP-A, histone H4, and DNA fragments, the researchers zeroed in on the minimalist core components of centromeric chromatin. This selective approach allowed them to capture the essence of centromere-specific nucleosome formation, free from the confounding presence of other histones.
.adsslot_7GCSd1vW3u{ width:728px !important; height:90px !important; }
@media (max-width:1199px) { .adsslot_7GCSd1vW3u{ width:468px !important; height:60px !important; } }
@media (max-width:767px) { .adsslot_7GCSd1vW3u{ width:320px !important; height:50px !important; } }
ADVERTISEMENT
Utilizing cryo-electron microscopy (cryo-EM), a cutting-edge technique that visualizes macromolecules at near-atomic resolutions, the team achieved an extraordinary 3.66 Ångström resolution of this subnucleosomal particle. The resultant structural details underscore the uniqueness of the CENP-A–H4 octasome, revealing DNA wrapping of approximately 120 base pairs—significantly less than the 145 base pairs bound in conventional nucleosomes. This reduced DNA length suggests a more compact or altered nucleosome conformation, one potentially optimized for facilitating kinetochore protein recruitment.
One of the striking features distinguishing the CENP-A–H4 octasome from canonical nucleosomes lies in the presence of four RG-loops derived from CENP-A histones. These RG-loops protrude as accessible scaffolds, providing twice the number of potential interaction sites compared to classical CENP-A nucleosomes that include histone H2A and H2B. This structural adaptation is hypothesized to underpin the recruitment and assembly of kinetochore components at the centromere, orchestrating the faithful segregation of chromosomes.
Moreover, the novel octasome exhibits a profound alteration in surface charge properties. Unlike canonical nucleosomes possessing an acidic patch— a negatively charged surface domain critical for anchoring histone methyltransferases and chromatin remodeling complexes— the CENP-A–H4 octasome lacks this feature. The absence of the acidic patch hints at fundamentally different modes of epigenetic regulation and chromatin dynamics within centromeric domains, potentially facilitating a unique chromatin landscape that differentiates it from conventional nucleosomal arrays.
These revelations are not merely structural curiosities; they bear significant biological implications. The precise mechanism by which the CENP-A–H4 octasome influences kinetochore assembly offers tantalizing insights into the causes of chromosomal missegregation—a hallmark of numerous genetic disorders and cancers. Aberrant expression levels of CENP-A have been implicated in mitotic errors, and understanding the molecular underpinnings of its arrangement could pave the way for novel therapeutic interventions targeting cell division anomalies.
Beyond expanding the molecular framework of centromere function, this discovery challenges long-standing dogmas regarding nucleosome composition and chromatin organization. The concept of a subnucleosome, devoid of certain histone pairs yet structurally stable and functionally significant, introduces new paradigms in chromatin biology. It raises important questions about how histone variants and nucleosome remodeling collaborate to sculpt the epigenetic landscape necessary for genome stability.
The adoption of the CENP-A–H4 octasome structure by eukaryotic cells may reflect an evolutionary adaptation to the mechanical and regulatory demands of chromosome segregation. The unique balance of DNA wrapping, histone composition, and exposed interaction loops suggests a finely tuned molecular machine evolved to meet the stringent fidelity requirements of mitosis.
Furthermore, this research underscores the power of cryo-EM in resolving delicate macromolecular architectures that have resisted traditional structural biology approaches. By visualizing nucleosomes at near-atomic resolution, the researchers provide an invaluable template for future studies aiming to dissect complex chromatin assemblies and their functional ramifications.
Intriguingly, these findings also broaden our understanding of epigenetic regulation at the centromere. By revealing differences in surface charge and histone subunit composition, the study suggests that chromatin remodelers and epigenetic modifiers may engage centromeric chromatin in ways previously unappreciated, underscoring the specialized nature of centromeric epigenetics.
In summary, the elucidation of the CENP-A–H4 octasome not only advances the frontier of molecular cell biology but also sets the stage for deeper exploration into the mechanistic foundations of faithful chromosome inheritance. This groundbreaking work from the Institute of Science Tokyo represents a critical leap toward deciphering the sophisticated choreography of proteins and DNA that preserves genomic integrity through generations of cells.
Subject of Research: Cells
Article Title: Cryo-EM Analysis of a Unique Subnucleosome Containing Centromere-Specific Histone Variant CENP-A
News Publication Date: 24-Mar-2025
Web References:
https://onlinelibrary.wiley.com/doi/10.1111/gtc.70016
http://dx.doi.org/10.1111/gtc.70016
Image Credits: Institute of Science Tokyo, Japan
Keywords: Molecular biology, Chromosome structure, Chromosomal abnormalities, Cancer cells, Cell division, Cell biology, Epigenetics, Histones, Mitosis, DNA, Cancer, Nucleosomes
Tags: CENP-A–H4 octasomecentromere function and biologycentromere molecular structurechromatin organization studieschromosome architecture researchgenetic material distributionhistone variants in centromereskinetochore assembly mechanismsmitosis and chromosome segregationmolecular characterization of centromeresnucleosome-like particlesprotein machineries in cell division