Credit: WANG Feng
A major challenge in neuroscience lies in understanding how molecular and circuit differences in the brain synergistically contribute to sex differences in behavioral phenotypes. Sex differences in the brain may arise from the identity, distribution and relative abundance of cell types.
The cannabinoid 1 receptor (CB1R), the main receptor for cannabinoids and endocannabinoids (eCB), is the most abundantly expressed G protein-coupled receptor in the brain and is highly conserved in animal evolution. However, whole-brain mapping of CB1R expression in adult male and female mice had not previously been described in detail.
Recently, a research team led by Dr. WANG Feng from the Shenzhen Institutes of Advanced Technology (SIAT) of the Chinese Academy of Sciences demonstrated that CB1R mRNA expression displays a sexually dimorphic pattern in several regions in the adult mouse brain.
According to their current study published in Journal of Comparative Neurology on January 29, a systemic mapping of CB1R mRNA expression was conducted and compared for the first time in male and female adult C57BL/6J mouse (a common inbred strain of laboratory mouse) brains.
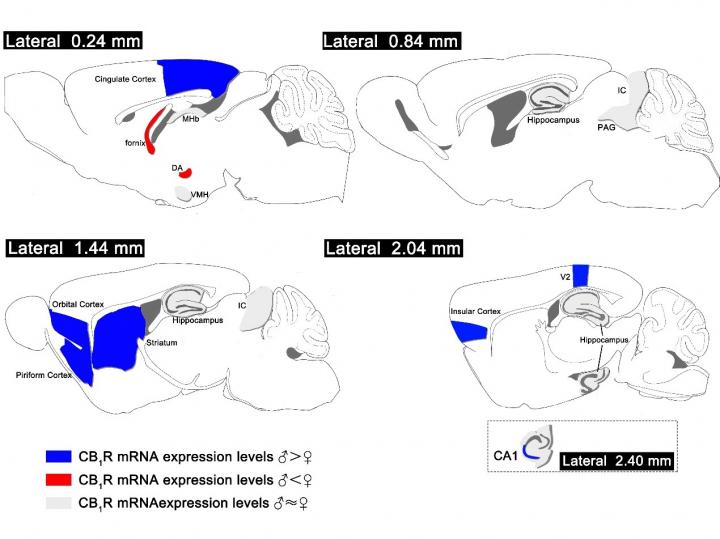
The researchers found that the neocortex, medial habenula, striatum, hippocampus and cerebellum were enriched with CB1R mRNA signals.
In the orbital cortex, insular cortex, cingulate cortex, piriform cortex, secondary visual cortex, caudate putamen and ventral hippocampus CA1, there were significantly more CB1R positive cells in males than in females. On the contrary, in the fornix and dorsal hypothalamus, CB1R positive cells were displayed more in females.
It is plausible that the difference in CB1R expression in the brain contributes to sex differences in various phenotypes. For example, the striatum, which harbors a greater amount of CB1R positive cells in males, has been widely recognized as the main affected region in Parkinson’s disease, which has a higher incidence in males. The dorsal hypothalamus, with a female-biased number of CB1R positive cells, is associated with maternal behaviors.
More interestingly, CB1R mRNA seems to fluctuate with the estrus cycle in female brain regions, including the medial amygdala, basolateral amygdala and parabrachial nucleus, which has not been reported before.
This study maps the localization of CB1R positive cells on a whole-brain scale in both genders for the first time. The findings provide a basis for understanding sexual dimorphism in physiological and pathological brain functions related to cannabinoids and endocannabinoids.
###
Media Contact
ZHANG Xiaomin
[email protected]
Original Source
http://english.
Related Journal Article
http://dx.