Credit: Berkeley Lab
Lithium Doesn’t Crack Under Pressure, It Transforms
by ALIYAH KOVNER
Using cutting-edge theoretical calculations performed at NERSC, researchers at Berkeley Lab’s Molecular Foundry have predicted fascinating new properties of lithium – a light alkali metal that has intrigued scientists for two decades with its remarkable diversity of physical states at high pressures.
“Under standard conditions, lithium is a simple metal that forms a textbook crystalline solid. However, scientists have shown that when you put a lithium crystal under pressure, the atomic structure changes and, somewhat counterintuitively, its conductivity drops, becoming less metallic,” said Stephanie Mack, a graduate student research assistant at Berkeley Lab and first author of the study, published in PNAS. “We’ve discovered it also becomes topological, with electronic properties similar to graphene.”
Topological materials are a recently discovered class of solids that display exotic properties, such as having insulating interiors yet highly conductive surfaces, even when deformed. They are exciting for potential applications in next-generation electronics and quantum information science. According to co-authors Sinéad Griffin and Jeff Neaton, lithium becomes topological at high but experimentally achievable pressures, comparable to one-quarter of the pressure at the Earth’s center.
Thus far, most experimentally verified topological materials contain heavy, potentially toxic, elements. “But we’ve predicted that lithium, ostensibly a simple metal, can also have these unique properties,” said Mack.
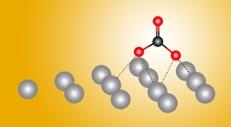
A ‘Silver Bullet’ for the Chemical Conversion of Carbon Dioxide
By Lori Tamura
Fossil fuels are the lifeblood of modern societies, but their increased use releases carbon dioxide, a climate-warming greenhouse gas, faster than plants can recycle it via photosynthesis.
Now, a powerful combination of experiment and theory has revealed atomic-level details about how silver helps transform carbon dioxide gas into a reusable form. The results, reported in the journal Nature Communications, will help in the design of more efficient metal catalysts.
“Before, people always thought that the process was the same on all metals,” said Berkeley Lab researcher Yifan Ye, one of the study’s authors. “But now, we have discovered that there are other options for reactions. This is new chemistry, and it’s a new reaction pathway.”
Metals such as silver facilitate the transformation (or “reduction”) of carbon dioxide into carbon monoxide (CO), which is used to synthesize other useful chemicals. The work revealed a surprising first step in this process that hadn’t been seen nor suggested before. Ultimately, the researchers hope to optimize carbon dioxide catalysis by using additives or metal alloys.
The work involved a close collaboration between theorists from Caltech and experimentalists from Berkeley Lab’s Advanced Light Source, working together under the umbrella of the Joint Center for Artificial Photosynthesis, a Department of Energy Energy Innovation Hub.
Read the full story here.
Media contact: Laurel Kellner, [email protected], 510-590-8034
Understanding Microbiomes for Advanced Wastewater Treatment and Reuse Systems
JULIE CHAO
Wastewater is treated by an activated sludge process in municipal wastewater treatment plants and returned to the environment for use. This treatment process has been used for over a century, and today represents the largest application of biotechnology in the world, yet there has been no effort to map the global activated sludge microbiome.
A study recently published in Nature Microbiology reports the first comprehensive, highly coordinated effort to examine the global diversity and biogeography of this microbiome. “The campaign involved 111 investigators who sampled 269 wastewater treatment plants in 86 cities in 23 countries on six continents,” said Jizhong Zhou, a professor of microbiology at the University of Oklahoma and Berkeley Lab adjunct senior scientist.
The researchers found a highly diverse activated microbiome, containing up to one billion microbial phylotypes comprised of novel species. “This expansive study is the first time that a systematic study of the hugely beneficial microbial communities involved in the biological treatment of daily wastewaters from communities around the world have been studied to understand their fundamental structure and function has been undertaken. It represents an important development in understanding and maintaining these crucial microbial communities,” said Lisa Alvarez-Cohen, a co-author and adjunct senior scientist at Berkeley Lab.
Read the full release from the University of Oklahoma here.
To learn more about Berkeley Lab’s water-energy research, visit the site of our Water-Energy Resilience Research Institute.
###
Media Contact
Laurel Kellner
[email protected]
Original Source
https:/




