In a pioneering stride toward conquering some of the most challenging cancers, researchers at Nagoya University in Japan, in collaboration with international partners, have developed a next-generation CAR-T cell therapy that shows remarkable promise in targeting and eradicating solid tumors. Published in the Journal for ImmunoTherapy of Cancer, this innovative approach harnesses the molecular target Eva1 (also known as MPZL2), a protein prevalently expressed on various malignant tumors, setting a new benchmark in the field of cancer immunotherapy traditionally limited to hematological malignancies.
CAR-T cell therapy—short for Chimeric Antigen Receptor T-cell therapy—revolutionized cancer treatment by genetically engineering patients’ own T cells to recognize and eliminate cancer cells. While these engineered cells have demonstrated unprecedented success in treating blood cancers like leukemia and lymphoma, solid tumors have remained recalcitrant due to their complex microenvironments and limited accessibility. Overcoming these barriers calls for refined and specialized CAR designs tailored to the unique biology of solid tumors.
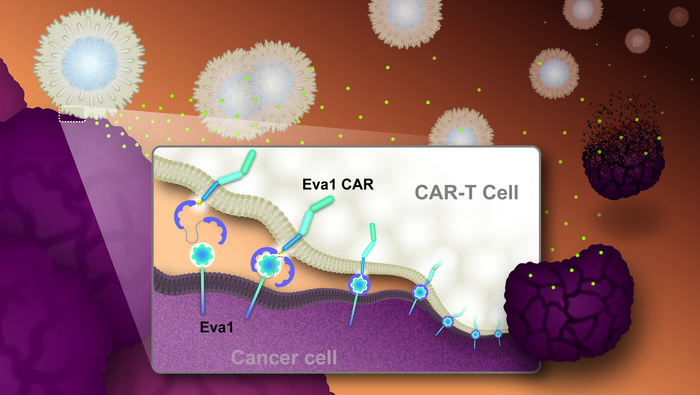
The Nagoya team singled out Eva1, a less explored but compelling antigen, given its unusually high expression on lung, pancreatic, and liver tumor cells, alongside relatively sparse distribution on normal tissues. This antigen’s small molecular footprint augurs well for enhanced immune cell engagement. Eva1’s diminutive size enables CAR-T cells to form stronger and more effective immunological synapses—critical junctions where immune cells and their targets physically connect, facilitating superior signaling that boosts T-cell activation and antitumor functions.
.adsslot_9G3ktqQEd1{ width:728px !important; height:90px !important; }
@media (max-width:1199px) { .adsslot_9G3ktqQEd1{ width:468px !important; height:60px !important; } }
@media (max-width:767px) { .adsslot_9G3ktqQEd1{ width:320px !important; height:50px !important; } }
ADVERTISEMENT
Central to their breakthrough was the intricate engineering of the CAR construct itself, focusing on two pivotal aspects: the spacer region and the intracellular domains. The spacer dictates the spatial configuration between the CAR-T cell and the tumor cell during contact, influencing the strength and duration of cell-to-cell interactions. Meanwhile, intracellular signaling domains modulate the activation state, persistence, and cytotoxic potency of the CAR-T cells. By creating sixteen variant CARs featuring combinations of humanized Eva1-binding antibodies, tailored spacer lengths, and distinct intracellular co-stimulatory motifs, the researchers identified ideal configurations that maximized therapeutic impact.
Humanization of the antibody fragment was crucial for clinical translation. Originally derived from mouse antibodies against Eva1, the binding domains were restructured to closely mimic human antibodies, minimizing the risk of adverse immune rejection when administered to patients. This refined design specifically increased affinity and selectivity for Eva1, reducing off-target effects and ensuring that the CAR-T cells preferentially recognize malignant, high-Eva1-expressing tumor cells.
Among the configurations tested, those employing a short spacer combined with co-stimulatory intracellular domains 4-1BB or a dual CD79A/CD40 module stood out. These constructs conferred superior expansion, cytokine secretion, and cytotoxic capabilities upon CAR-T cells, culminating in highly effective elimination of tumors in murine models that mimic human lung and pancreatic cancers. Such preclinical success underscores a potential leap forward in tackling solid tumors, which have been notoriously refractory to existing immunotherapies.
Safety, a paramount concern in CAR-T therapy, was rigorously evaluated given that Eva1 is not completely tumor-specific and is also present in low amounts on normal monocytes, a subset of white blood cells. Encouragingly, the engineered CAR-T cells demonstrated exquisite sensitivity to antigen density, activating robustly only upon encountering cells with high Eva1 expression typical of cancer cells, while largely sparing normal monocytes. This on-target, off-tumor discrimination signifies a promising safety profile, essential to minimize collateral damage and treatment-related toxicities in future clinical applications.
The sophisticated immune synapse formation observed with Eva1CAR-T cells may hold the key to their enhanced efficacy. Due to Eva1’s molecular structure and size, the engineered T cells can establish more intimate and stable physical contacts with cancer cells, reinforcing sustained T-cell receptor signaling, cytokine production, and proliferative responses. These features collectively drive more potent and durable antitumor immunity, overcoming the limitations seen in earlier CAR-T designs targeting bulkier or less accessible antigens.
Dr. Seitaro Terakura, lead investigator from Nagoya University’s Graduate School of Medicine, emphasized the clinical significance of these findings. He noted that the strategy offers a tangible pathway for treating solid tumors that have thus far evaded effective immune-based therapies. Tumors of priority include lung, pancreatic, and liver cancers—malignancies responsible for significant global mortality, often diagnosed at advanced stages with poor prognosis under current therapeutic regimes.
The team is now poised to translate this promising preclinical research into human trials. Before this can occur, thorough safety assessments are underway using mouse models engineered to express mouse Eva1. Developing a murine Eva1-specific CAR-T allows detailed toxicity profiling, verifying that the therapy does not induce deleterious damage to normal tissue expressing basal levels of Eva1. Successful demonstration of safety will pave the way for pivotal clinical trials in patients, moving closer to the ultimate goal of offering a lifesaving intervention.
Looking forward, the researchers plan to collaborate with biotech and pharmaceutical partners to advance clinical development. The optimization framework established here—combining antigen selection, CAR spacer engineering, and intracellular co-stimulatory domain tuning—may also provide a blueprint for developing therapies against other challenging tumor antigens. This modular and rational design paradigm promises to expand the arsenal of effective, safe, and targeted CAR-T cell therapies for solid malignancies.
As this approach transitions from bench to bedside, it promises enormous implications not only for patient outcomes but also for the broader field of cancer immunotherapy. Harnessing the immune system’s power with precision-engineered cellular therapies signals a new dawn where even the most intractable cancers might be conquered with minimal toxicity and maximal clinical benefit.
The success of Eva1-targeting CAR-T cells embodies the fusion of cutting-edge molecular engineering, immunological insight, and translational ambition. It underscores how targeted molecular design can overcome biological hurdles previously thought insurmountable, offering hope for more effective treatments against the world’s deadliest cancers in the near future.
Subject of Research: Animals
Article Title: Development and optimization of Eva1 (MPZL2) targeting chimeric antigen receptor T cells
News Publication Date: 7-May-2025
Web References: Journal for ImmunoTherapy of Cancer, DOI: 10.1136/jitc-2024-009825
Image Credits: Keiko Itano, Nagoya University
Keywords: Cancer immunotherapy, Immune cells, Antibodies, Antigens, Immune response, Cancer cells, Cancer research, Liver cancer, Lung cancer, Pancreatic cancer, Adoptive T cell therapy
Tags: CAR T cell therapy advancementscollaboration in cancer researchenhancing immune response in cancerEva1 antigen and cancer treatmentgenetic engineering in cancer cellsinnovative cancer immunotherapy strategiesmolecular targets in oncologyNagoya University cancer researchnext-generation cancer therapiesovercoming challenges in solid tumor therapypersonalized cancer treatment approachestargeting solid tumors with CAR-T cells