Researchers show that procedures used to contain Candida auris infection in an animal facility can potentially be applied to hospitals and nursing homes to limit its spread
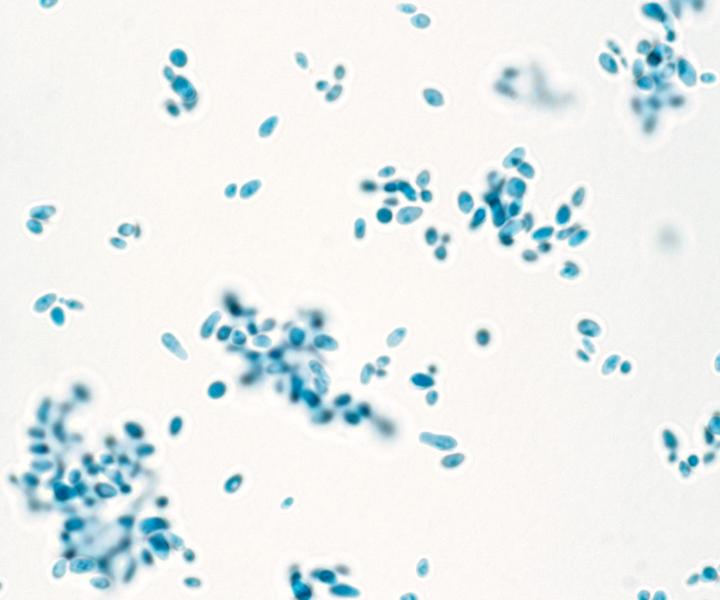
Credit: Dr. Fernando Torres-Velez, Wadsworth Center.
Researchers show that procedures used to contain Candida auris infection in an animal facility can potentially be applied to hospitals and nursing homes to limit its spread.
A new study published in the Journal of Occupational and Environmental Health has established protocols for containing the drug resistant Candida auris (C. auris ) in an animal facility, and by doing so, has identified four simple rules that can potentially be adopted by healthcare facilities to limit exposure to staff and patients. The study found that their double personal protective equipment (PPE), work ‘buddy’ system, disinfection and biomonitoring protocols were effective at containing high levels of C. auris infection within their animal facility, even six months after their experiments.
This study was a collaborative effort between a team of researchers led by Dr. Magdia De Jesus, Assistant Professor at the University at Albany, State University of New York, and Research Scientist at the Wadsworth Center, New York State Department of Health; biosafety officers and the Clinical Mycology laboratory at the Wadsworth Center.
“C. auris is a drug resistant, transmissible pathogen, and because it is newly emerging, better animal models are needed to understand the pathogenesis of the disease,” said Dr. De Jesus, who supervised the study. “C. auris is a serious drug resistant organism. Working together we can help minimise its spread in healthcare settings and prevent skin colonization.”
Current mouse models use very high inoculums of the C. auris pathogen, but no-one has tested the risk of exposure for people working in these environments. This study was intended to develop a standard operating procedure to keep researchers and animal care staff safe.
The researchers inoculated mice with 105 to 108 C. auris cells and set up a workflow of protocols to contain the infection within the animal holding and procedure rooms during their experiments. The workflow and protocols were set up around four main elements: PPE, a buddy system, disinfection and biomonitoring of surfaces.
Before entering the animal holding and procedure rooms, staff donned a second layer of booties, gloves and gowns, which were later removed and placed in biohazard bins before exiting the rooms. Handling of infected cages and equipment was restricted to biosafety cabinets where a buddy system was implemented so that one person handed clean cages and supplies to a second person working inside the contaminated biosafety cabinet. This system-controlled workflow from clearly defined ‘clean’ to ‘dirty’ areas and allowed workers to monitor each other to ensure proper procedures were followed. Surfaces and equipment that came in contact with infected mice or tissues were treated with a strict disinfection protocol of 10% bleach followed (after five minutes) by 70% ethanol. The effectiveness of the workflow and protocols were continually monitored using swab testing on surfaces suspected to be contaminated, and as a second measure, Sabbaroud dextrose plates were placed inside the biosafety cabinet and on the floor underneath to determine whether C. auris was aerosolised within the cabinet or whether any debris contaminated the floor.
The researchers found that possible contamination came from direct contact with the infected mice or tissues but not from aerosolisation.
Despite the mice being clearly infected, all the surfaces researchers tested were negative for C. auris infection This told them that their procedures can contain the infection, even six to 12 months after more than 20 experiments were conducted with high inoculums.
Although their research was geared towards containing C. auris in an animal facility, the researchers argue that their procedures are applicable to other healthcare environments.
The research team identified four simple rules which they believe can prevent the spread of C. auris in most situations. Facilities should develop a PPE system where healthcare workers wear double gloves, gowns and booties, with appropriate receptacles placed inside and outside a patient’s room for donning and removal of PPE; a 10% bleach, for five-minutes, followed by 70% ethanol disinfection protocol; a buddy system where one staff member watches that the patient-care worker is following procedures and to hand over supplies; and finally, regular swab-testing to ensure surfaces are cleaned effectively..
Overall, the study found that these four steps (PPE, buddy system, disinfection, biomonitoring) contained the high levels of C. auris infection during their mouse model testing, and these procedures are potentially transferable to hospitals and nursing homes to protect staff and patients and limit further spread of the infection.
###
Peer-reviewed; Experimental Study; Animals
About Taylor & Francis Group
Taylor & Francis Group partners with researchers, scholarly societies, universities and libraries worldwide to bring knowledge to life. As one of the world’s leading publishers of scholarly journals, books, ebooks and reference works our content spans all areas of Humanities, Social Sciences, Behavioural Sciences, Science, Technology and Medicine.
From our network of offices in Oxford, New York, Philadelphia, Boca Raton, Boston, Melbourne, Singapore, Beijing, Tokyo, Stockholm, New Delhi and Cape Town, Taylor & Francis staff provide local expertise and support to our editors, societies and authors and tailored, efficient customer service to our library colleagues.
Media Contact
Saskia Kovandzich
[email protected]
Related Journal Article
http://dx.