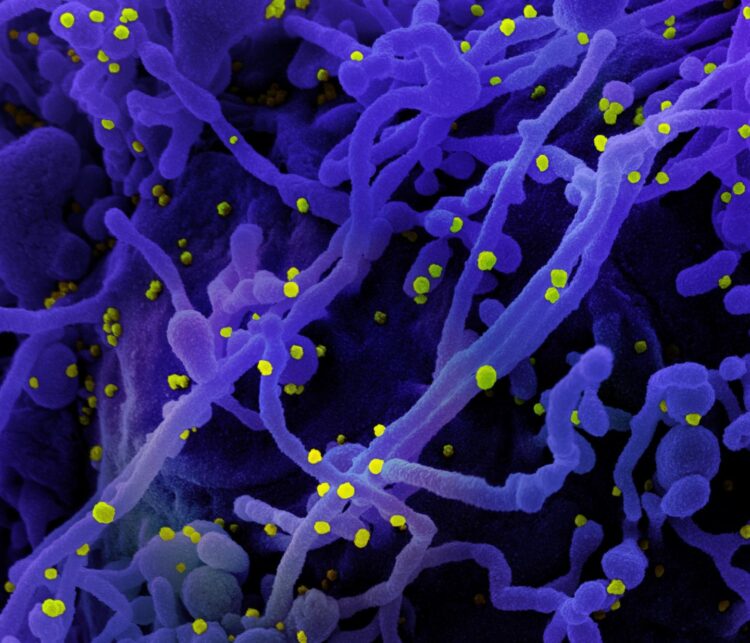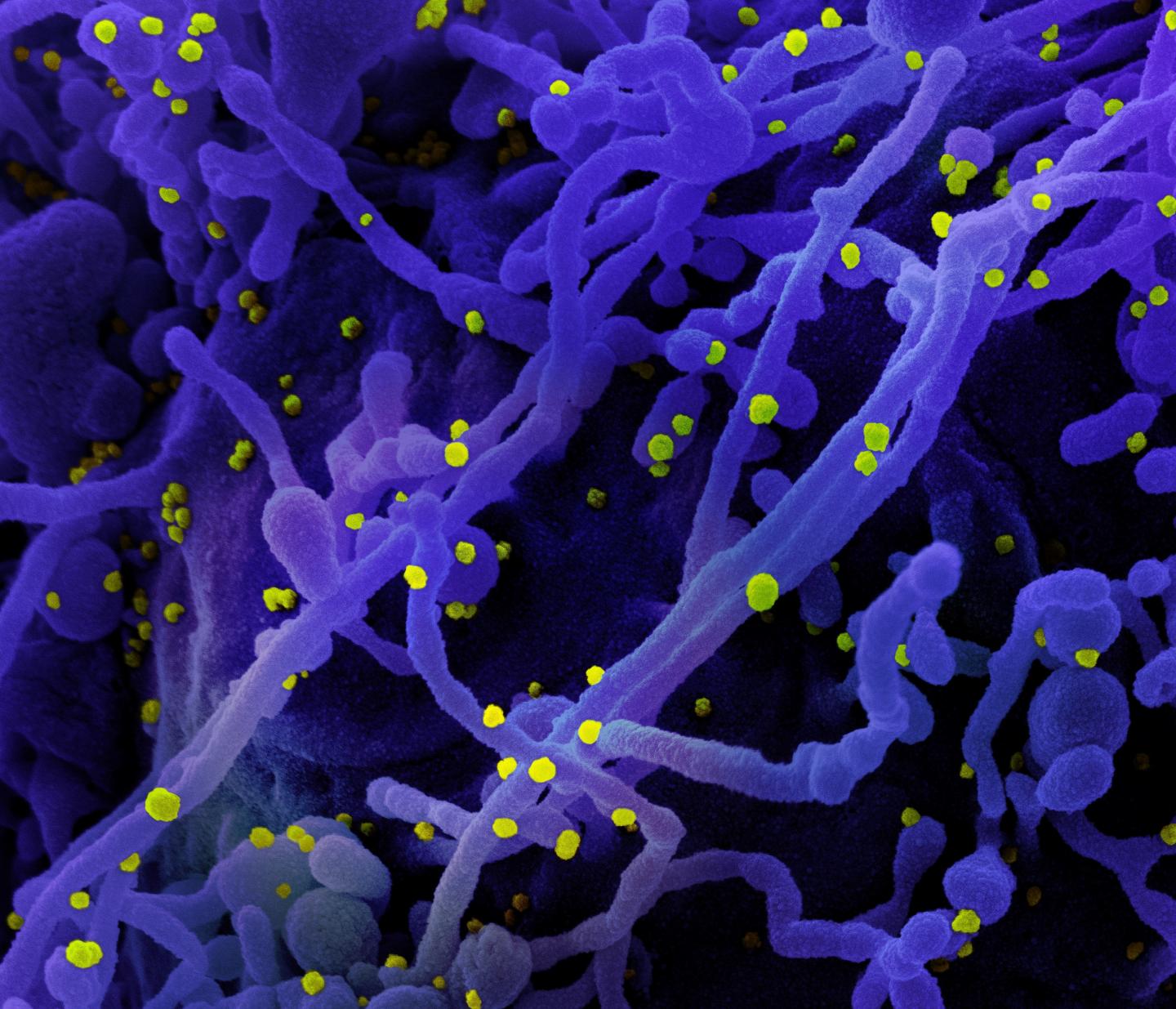
Credit: NIAID
WHAT:
NIH investigators and colleagues have discovered that when the immune system first responds to infectious agents such as viruses or bacteria, a natural brake on the response prevents overactivation. Their new study in mBio describes this brake and the way pathogens such as SARS-CoV-2, the virus that causes COVID-19, turn it on. Their finding provides a potential target for an immunotherapy that might be applied to a wide range of infectious diseases.
When a cell senses an infectious agent with molecules called pathogen recognition receptors, part of its response is to increase cell surface expression of a molecule called CD47, otherwise known as the “don’t eat me” signal. Increased CD47 expression dampens the ability of cells called macrophages, the immune system’s first responders, to engulf infected cells and further stimulate the immune response. Upregulation of CD47 on cells was observed for diverse types of infections including those caused by mouse retroviruses, lymphocytic choriomeningitis virus, LaCrosse virus, SARS CoV-2, and by the bacteria Borrelia burgdorferi and Salmonella enterica typhi.
By blocking CD47-mediated signaling with antibodies in mice infected with lymphocytic choriomeningitis virus, the authors demonstrated they could enhance the speed of pathogen clearance. Furthermore, knocking out the CD47 gene in mice improved their ability to control M. tuberculosis infections and significantly prolonged their survival. In addition, retrospective studies of cells and plasma from people infected with hepatitis C virus indicated that humans also upregulate CD47. In these studies, inflammatory cytokine stimuli and direct infection both promoted increased CD47 expression.
This highly collaborative research project involved 14 different institutions and was led by scientists from NIH’s National Institute of Allergy and Infectious Diseases in Hamilton, Montana, and Stanford University in Stanford, California. The findings open the possibility of using CD47 blockade as a new immunotherapeutic to treat a wide range of different infections. “There may be circumstances where host responses need boosting and CD47 represents a novel target for host-directed therapies in such cases,” the scientists write, mentioning SARS-CoV-2, HIV, HPV and Ebola virus among several possibilities.
###
ARTICLE:
M Caspi Tal et al. Upregulation of CD47 is a host checkpoint response to pathogen recognition. mBio DOI: 10.1128/mBio.01293-20 (2020).
WHO:
Kim J. Hasenkrug, Ph.D., Chief of NIAID’s Retroviral Immunology Section, is available for comment.
CONTACT:
To schedule interviews, please contact Ken Pekoc, (301) 402-1663, [email protected].
NIAID conducts and supports research–at NIH, throughout the United States, and worldwide–to study the causes of infectious and immune-mediated diseases, and to develop better means of preventing, diagnosing and treating these illnesses. News releases, fact sheets and other NIAID-related materials are available on the NIAID website.
About the National Institutes of Health (NIH): NIH, the nation’s medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit http://www.
Media Contact
Ken Pekoc
[email protected]
Original Source
https:/
Related Journal Article
http://dx.