In a groundbreaking investigation into the molecular mechanisms governing osteosarcoma progression and treatment response, scientists have unveiled the paradoxical role of microRNA-767-5p (miR-767-5p) in modulating both metastasis and therapeutic sensitivity. This dual-function microRNA appears to simultaneously accelerate the spread of osteosarcoma cells while rendering them more vulnerable to chemotherapy and radiotherapy—a discovery that challenges conventional understanding of tumor biology and opens novel avenues for targeted cancer therapies.
Osteosarcoma (OS) is the most common primary malignant bone tumor, predominantly affecting children and young adults. Despite advances in surgical techniques and chemotherapeutic regimens, metastatic osteosarcoma remains a significant clinical problem, often culminating in poor prognosis and high mortality rates. Researchers have directed their attention to microRNAs—small, non-coding RNA molecules that regulate gene expression—as they increasingly recognize their critical contributions to cancer pathogenesis, progression, and treatment resistance.
The study, published in BMC Cancer, focused on deciphering the role of miR-767-5p in OS. Analysis of patient specimens revealed that miR-767-5p is notably upregulated in individuals with metastatic OS compared to healthy controls, suggesting its involvement in malignant progression. Moreover, the elevated expression of miR-767-5p showed a robust negative correlation with overall patient survival, indicating its potential as a prognostic biomarker in clinical settings.
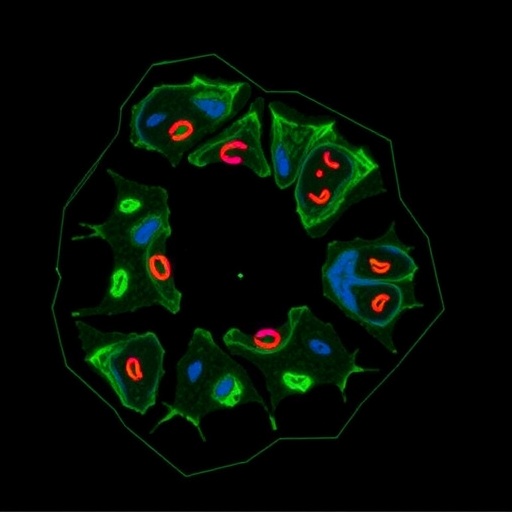
To unravel the functional implications of miR-767-5p modulation, the research team employed well-established human osteosarcoma cell lines, U2OS and 143B, conducting a series of in vitro assays including Cell Counting Kit-8 (CCK-8), transwell migration and invasion, and colony formation assays. These experiments demonstrated that overexpression of miR-767-5p significantly enhanced the invasive and migratory capabilities of OS cells but intriguingly did not affect their proliferation rates. Conversely, knockdown of miR-767-5p curtailed these metastatic traits, underscoring its regulatory influence on tumor cell dissemination.
Extending their findings to an in vivo context, the investigators utilized a tumor xenograft model, which recapitulated the pro-metastatic effects of miR-767-5p overexpression observed in vitro. This convergence of cellular and animal data reinforced the conclusion that miR-767-5p is a potent driver of OS metastasis. However, what sets this study apart is the unexpected revelation that miR-767-5p simultaneously sensitizes OS cells to chemotherapy agents and ionizing radiation, two cornerstone modalities in osteosarcoma treatment.
By subjecting U2OS and 143B cells to chemotherapeutic drugs and radiotherapy, the researchers observed a pronounced enhancement in treatment efficacy upon miR-767-5p overexpression. This counterintuitive phenomenon suggests a complex regulatory network whereby miR-767-5p promotes malignancy while impairing cellular resistance mechanisms. Such duality presents a clinically exploitable vulnerability, potentially allowing oncologists to tailor treatment strategies that leverage this microRNA’s sensitizing effects while managing metastatic risk.
To elucidate the molecular underpinnings of miR-767-5p’s dual role, the team integrated bioinformatics-based target gene prediction with RNA-sequencing analyses and patient survival data. This comprehensive approach pinpointed the aryl hydrocarbon receptor (AHR), a ligand-activated transcription factor involved in xenobiotic metabolism and cellular stress responses, as a key downstream target of miR-767-5p. Luciferase reporter assays verified that miR-767-5p directly interacts with the 3’-untranslated region (3’-UTR) of AHR mRNA, modulating its expression post-transcriptionally.
Further experimental validation revealed that enforced expression of AHR in OS cells antagonized the phenotypic effects of miR-767-5p, suppressing cell invasion and migration while diminishing sensitivity to chemo- and radiotherapy. These findings establish a functional axis in which miR-767-5p downregulates AHR to facilitate metastasis but concurrently impair resistance pathways, thereby increasing therapeutic susceptibility.
Understanding the miR-767-5p/AHR regulatory pathway enriches the current knowledge landscape surrounding osteosarcoma biology, highlighting the intricate balance between tumor aggressiveness and treatment responsiveness. This insight not only elucidates part of the molecular circuitry driving OS progression but also proposes potential intervention points for pharmacological modulation.
The clinical implications of this study are profound. Targeting miR-767-5p or its interaction with AHR could offer a dual-pronged strategy, simultaneously impeding metastatic dissemination and enhancing the efficacy of existing treatment regimens. Such approaches might include the development of miRNA mimics, inhibitors, or small molecules designed to modulate this pathway with precision, potentially improving patient outcomes in a disease notorious for its therapeutic challenges.
Moreover, given the negative correlation between miR-767-5p levels and patient survival, this microRNA could serve as a valuable biomarker for stratifying osteosarcoma patients based on metastatic risk and expected treatment response. Incorporating miR-767-5p profiling into routine diagnostics could thus guide personalized medicine, tailoring interventions to maximize benefit while mitigating adverse effects.
This study serves as a clarion call for further exploration into the complex roles of microRNAs in creating tumor vulnerabilities that can be therapeutically exploited. The paradoxical activities of miR-767-5p exemplify the multifaceted nature of cancer regulators and underscore the necessity of integrative, multidisciplinary research approaches.
Future research trajectories may involve dissecting the downstream signaling cascades influenced by AHR suppression, assessing how these molecular changes impact DNA damage response pathways and apoptotic processes during chemotherapy and radiotherapy. Additionally, exploring the role of the tumor microenvironment and immune modulation in the context of miR-767-5p expression may shed light on holistic interactions influencing OS pathobiology.
Continued investigation using patient-derived xenograft models and clinical trials will be essential to translate these molecular insights into effective treatment modalities. Ultimately, the goal is to harness the intricate molecular interplay revealed by this study, converting biological paradoxes into therapeutic triumphs against osteosarcoma.
As osteosarcoma remains a formidable clinical challenge, discoveries such as the miR-767-5p/AHR axis illuminate previously uncharted territories where molecular intervention can reshape the landscape of cancer care. This dual role of miR-767-5p—catalyzing metastasis while amplifying treatment sensitivity—redefines traditional paradigms and sets a precedent for nuanced, mechanism-driven therapeutic innovation.
Subject of Research: The study investigates the role of microRNA-767-5p (miR-767-5p) in osteosarcoma progression, metastasis, and sensitivity to chemotherapy and radiotherapy.
Article Title: MicroRNA-767-5p promotes metastasis but improves chemotherapeutic and radiotherapeutic sensitivity of osteosarcoma.
Article References:
Luo, X., Dai, X., Wei, Q. et al. MicroRNA-767-5p promotes metastasis but improves chemotherapeutic and radiotherapeutic sensitivity of osteosarcoma. BMC Cancer 25, 702 (2025). https://doi.org/10.1186/s12885-025-14114-y
Image Credits: Scienmag.com
DOI: https://doi.org/10.1186/s12885-025-14114-y
Tags: chemotherapy sensitivity in osteosarcomadual-function microRNA in cancermechanisms of tumor metastasismetastasis and cancer progressionmicroRNA as cancer biomarkersmicroRNA-767-5p role in osteosarcomanon-coding RNA in cancer researchnovel targeted cancer therapiespediatric osteosarcoma clinical challengesprognostic biomarkers for osteosarcomaradiotherapy response in bone tumorstherapeutic response in osteosarcoma