This large US study found that 1 in 5 patients was prescribed opioids after having a pacemaker or similar device implanted and 80 percent had never taken opioids before, report investigators in HeartRhythm
Credit: Mayo Foundation for Medical Education and Research.
Philadelphia, October 21, 2019 – One in five patients is prescribed opioids after having a pacemaker or similar device implanted, according to a large US study conducted at Mayo Clinic published in HeartRhythm, the official journal of the Heart Rhythm Society and the Cardiac Electrophysiology Society published by Elsevier. Eighty percent of patients who were prescribed opioids had never taken them before. Investigators stress the importance of improving postoperative pain management following cardiac device procedures to reduce use of prescription opioids.
Prescription opioids are a major contributor to the opioid epidemic in the US and are linked to prescription opioid abuse, addiction, and deaths due to overdose. More people now die due to prescription opioid overdose than from cocaine and heroin.
“Opioid prescription following cardiac implantable electronic device procedures (CIED) has not previously been studied,” explained lead investigator Siva K. Mulpuru, MD, Mayo Clinic Rochester, Rochester, MN, USA. “The main aim of our study was therefore to gain a better understanding of opioid prescription patterns following CIED procedures. Secondary objectives were to understand various factors associated with opioid prescriptions and to evaluate continued opioid prescription rates (refills) after initial surgery.”
Investigators performed a retrospective cohort study of all patients undergoing device procedures including implantation of pacemakers, cardioverter defibrillators, generator changes, and lead extractions at the Mayo Clinic Enterprise Heart Rhythm Practice. The study included patients at the three academic campuses in Rochester, MN; Phoenix, AZ; and Jacksonville, FL. More than 16,500 adult patients who underwent CIED procedures and were discharged between January 1, 2010 and March 30, 2018 were included in the study. Procedures were categorized into new implant, generator change, device upgrade, lead revision or replacement, and subcutaneous implantable cardiac defibrillator (S-ICD) procedures. The investigators assessed how frequently opioids were prescribed after the procedure, and how frequently patients who were prescribed opioids continued to refill their opioid prescriptions.
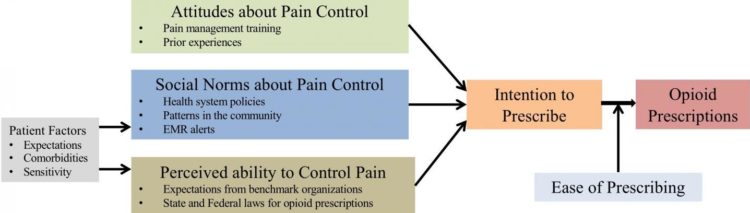
Opioids were prescribed to about 20 percent of the patients of whom 80 percent were “opioid naïve” (e.g., they had never taken opioids before). Among the opioid naïve patients who received opioids, more than nine percent subsequently refilled their prescription. Close to 40 percent of these patients received more than 200 oral morphine equivalents on prescription. The investigators concluded that several factors influence the decision to prescribe opioids, such as the healthcare provider’s pain management training and prior experiences; patients’ expectations of pain control; underlying comorbidities; and personal sensitivity to pain.
“The explanation for this high number of opioid prescriptions could be due to various reasons including provider factors, patient factors, and social norms,” noted first author Justin Z. Lee, MD, Mayo Clinic, Rochester, MN, USA. “We need to focus more on alternative pain management strategies to reduce the use of opioids after device procedures.”
“There is a clear need for more studies and awareness of perioperative pain management after device procedures to reduce postoperative pain and postoperative opioid prescriptions,” added co-author Abhishek Deshmukh, MBBS, Mayo Clinic, Rochester, MN, USA. “This ranges from various intraprocedural techniques to reduce postoperative pain to considerations of using a combination of scheduled nonopioid medications for a better nonopioid pain control strategy.”
###
Media Contact
Jane Grochowski
[email protected]
406-542-8397
Related Journal Article
http://dx.