In a groundbreaking advancement for precision oncology, researchers from Mayo Clinic and Stanford Medicine have unveiled an innovative blood test designed to decode the intricate ecosystem surrounding cancer cells within the body. This new approach, which delves far deeper than prior liquid biopsy techniques, offers oncologists an unprecedented window into the tumor microenvironment, enabling more accurate predictions regarding patient responses to immunotherapy. Published in the prestigious journal Nature, this study represents a monumental leap forward in personalized cancer treatment, potentially reshaping clinical decision-making across various cancer types.
Historically, liquid biopsies have focused predominantly on isolating and analyzing tumor cells circulating in the blood or tumor-derived DNA fragments. While such methods provided useful genetic insights, they largely overlooked the tumor’s complex microenvironment — the milieu of noncancerous cells, immune components, and stromal elements that significantly influence how tumors grow and respond to treatment. By shifting attention from tumor cells alone to the entire tumor neighborhood, this research offers a paradigm shift. It employs sophisticated molecular profiling to understand the cellular architecture and interactions that govern tumor behavior and immune response.
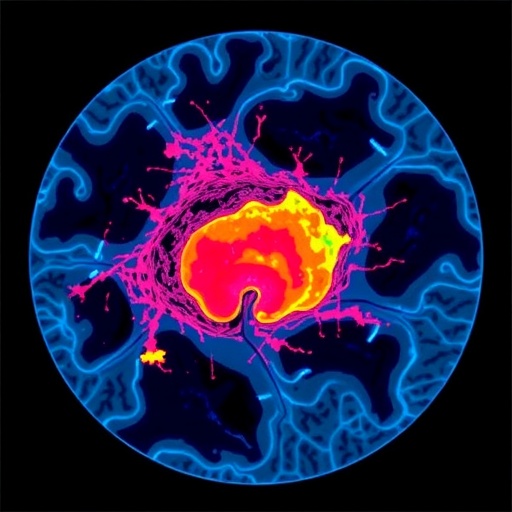
Central to this breakthrough is the application of spatial transcriptomics, a cutting-edge technique enabling scientists to map gene expression within the physical context of tissue architecture. Through detailed analysis of tumor specimens across multiple cancer types, researchers identified nine unique “spatial ecotypes” — distinctive cellular neighborhoods characterized by specific compositions of immune and stromal cells. These ecotypes were not random but spatially situated, with some residing at the tumor’s invasive edge adjoining healthy tissue, while others appeared deep within the tumor core. This spatial organization provides crucial insights into tumor biology and therapeutic vulnerability.
Recognizing the transformative potential of these findings, the team sought to extend spatial profiling beyond invasive tumor biopsies to a simple blood test. To achieve this, they partnered with experts in biomedical data science at Stanford Medicine who developed an artificial intelligence (AI) framework capable of interpreting methylation patterns on circulating tumor-derived cell-free DNA (cfDNA). DNA methylation—chemical tags regulating gene expression—serves as a fingerprint of the cellular origin and state. By decoding these methylation signatures, the AI model can infer the presence and proportions of the distinct spatial ecotypes circulating in the bloodstream, thus producing a dynamic portrait of the tumor microenvironment without the need for surgical sampling.
This noninvasive liquid biopsy not only profiles tumor ecologies with remarkable precision but also reveals critical correlations between specific ecotypes and patient outcomes. In extensive clinical validation involving over 1,300 individuals with malignancies such as melanoma, lung, bladder, and gastric cancers, certain spatial ecotypes strongly predicted who would benefit from immunotherapy. Patients whose tumors exhibited immune-rich ecotypes demonstrated markedly improved survival and response rates, whereas those with ecotypes associated with immune suppression or stromal barriers tended to resist therapy and have poorer prognoses. Intriguingly, this spatial ecotyping outperformed traditional biomarkers—such as tumor mutation burden or PD-L1 expression—in forecasting therapeutic success.
The clinical implications of this innovation are profound. Immunotherapies, while revolutionary, do not universally benefit all patients and often come with costs of significant toxicity and high expense. The ability to anticipate immunotherapy responsiveness through a blood test empowers oncologists to tailor treatments more effectively, sparing nonresponders from unnecessary side effects and allowing them to pursue alternate therapies sooner. Essentially, the test serves as a compass guiding more personalized, strategic treatment choices, improving both patient quality of life and survival outcomes.
Beyond initial treatment decisions, this novel blood test offers the potential for real-time monitoring of tumor evolution during therapy. Because it captures dynamic shifts in the tumor microenvironment’s cellular neighborhoods, oncologists can detect early signs of resistance or remission well before anatomical changes become visible through imaging techniques. This longitudinal insight may facilitate timely treatment modifications, optimizing therapeutic efficacy as the tumor adapts or responds over time.
While the research focus thus far has been on challenging cancers like melanoma, lung, and bladder cancer, the technology’s scope is promisingly broad. Early data suggest its utility in predicting complete responses to antibody drug conjugate (ADC)-based combination regimens, signaling a versatile tool that can decode treatment responses across multiple therapeutic modalities. Moreover, the approach’s principle—combining spatial transcriptomics and methylation-aware AI-driven liquid biopsy—holds promise beyond oncology, potentially deciphering complex pathologies in autoimmune diseases, infections, and other conditions where tissue microenvironments critically impact health.
The discovery unveiled by Dr. Aadel Chaudhuri and colleagues effectively opens a new window into biological complexity that was previously invisible through minimally invasive means. By tracing the tumor microenvironment’s spatial ecotypes via blood, clinicians and researchers alike gain access to a “geographic” map of the tumor’s cellular neighborhood, informing crucial decisions that may prevent overtreatment, identify therapeutic resistance early, and better personalize patient care pathways.
This research has already catalyzed patent filings and garnered commercial interest, signaling the translational potential of spatial ecotype profiling in oncology diagnostics. As ongoing studies aim to validate the assay in larger cohorts and refine its predictive algorithms, the eventual integration into routine clinical workflows may well redefine cancer management over the coming decade, making personalized immunotherapy selection as simple as a blood draw.
Ultimately, this pioneering liquid biopsy test exemplifies the power of combining molecular biology, spatial analytics, and artificial intelligence to illuminate the hidden landscapes of disease. As Dr. Chaudhuri emphasizes, this is just the beginning of harnessing complex biological environments noninvasively, with profound implications not only for cancer therapy but for broadening our understanding of multifaceted disease processes in humans.
Subject of Research: Noninvasive tumor microenvironment profiling and immunotherapy response prediction through liquid biopsy.
Article Title: Non-invasive profiling of the tumour microenvironment with spatial ecotypes
News Publication Date: 6-May-2026
Web References:
– Mayo Clinic News Network: https://newsnetwork.mayoclinic.org
– Nature Article: https://www.nature.com/articles/s41586-026-10452-4
References:
Chaudhuri, A., Newman, A., et al. Non-invasive profiling of the tumour microenvironment with spatial ecotypes. Nature. 2026; DOI:10.1038/s41586-026-10452-4.
Keywords:
liquid biopsy, tumor microenvironment, spatial transcriptomics, methylation profiling, artificial intelligence, immunotherapy, cancer biomarker, cell-free DNA, precision oncology, tumor spatial ecotypes, treatment response prediction, noninvasive diagnostics
Tags: cancer biomarker discoveryimmune microenvironment mappingimmunotherapy response predictionliquid biopsy advancementsliquid biopsy tumor ecosystemMayo Clinic Stanford cancer researchmolecular profiling of tumorspersonalized cancer treatmentprecision oncology blood testspatial transcriptomics in cancertumor microenvironment analysistumor neighborhood profiling