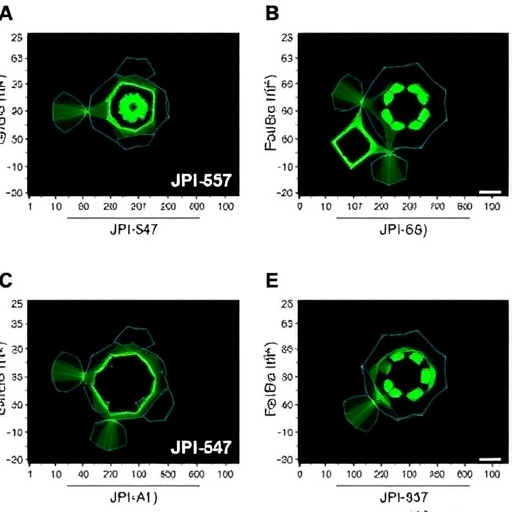
In a groundbreaking advance in the fight against hereditary breast and ovarian cancers, a newly developed molecule named JPI-547 has emerged as a potent contender, signaling a potential paradigm shift in targeted oncologic therapies. This innovative compound functions as a dual inhibitor, simultaneously targeting PARP1/2 and tankyrase enzymes, which marks a significant evolution from the first-generation PARP inhibitors that have dominated treatment protocols until now. As research deepens, JPI-547’s dual-action mechanism could rewrite the therapeutic playbook for challenging BRCA1/2-mutated tumors, promising enhanced efficacy and potentially superior clinical outcomes.
The heart of this breakthrough lies within the molecular sabotage of cancer’s DNA repair machinery. PARP1 and PARP2 enzymes play a critical role in single-strand DNA break repair via the base excision repair pathway. Tumor cells deficient in BRCA1 or BRCA2 genes already suffer from impaired homologous recombination, a high-fidelity DNA repair process. By inhibiting PARP enzymes, this vulnerability is exploited, leading to accumulated DNA damage and, ultimately, selective cancer cell death. However, first-generation PARP inhibitors, while effective, have clinical limitations, including resistance development and suboptimal therapeutic windows. JPI-547’s innovative dual inhibition of both PARP1/2 and tankyrase, a member of the poly(ADP-ribose) polymerase family involved in Wnt/β-catenin signaling, introduces a multi-pronged assault that could circumvent these challenges.
The addition of tankyrase inhibition is particularly noteworthy given the enzyme’s emerging role in tumor progression and cellular immortality. Tankyrase modulates key pathways linked to cellular proliferation and survival, primarily through its regulation of the Wnt/β-catenin pathway, integral in many cancers. By concurrently targeting tankyrase, JPI-547 disrupts both DNA repair and proliferative signaling, potentially undermining tumor resilience mechanisms that often limit the efficacy of conventional PARP inhibitors. This strategy signifies a sophisticated understanding of cancer biology, where multitargeted interventions are poised to transcend the limiting bottlenecks of monotherapies.
Preclinical investigations have been comprehensive and revealing. Using BRCA1/2-mutated cancer models, Kang, Katuwal, Ghosh, and colleagues systematically demonstrated that JPI-547 outperforms existing PARP inhibitors in suppressing tumor growth. These in vitro and in vivo models, employing patient-derived xenografts and genetically engineered cancer cells, highlight the compound’s capacity to induce DNA damage accumulation beyond what first-generation inhibitors accomplish. The synergy between impaired homologous recombination due to BRCA mutations and the compound’s dual blockade generates a synthetic lethality effect of pronounced magnitude, heralding superior anti-tumor activity.
Pharmacokinetic profiling of JPI-547 reveals favorable characteristics that increase its therapeutic appeal. Not only does it exhibit enhanced bioavailability, but it also maintains a longer half-life compared to its predecessors, potentially allowing for sustained target engagement with reduced dosing frequency. This could translate to fewer adverse effects and improved patient compliance, factors crucial in chronic cancer therapies. Importantly, the compound’s selective inhibition limits off-target toxicity, a common concern in poly(ADP-ribose) polymerase-targeting agents.
Molecular analyses further underscore how JPI-547 modulates critical signaling networks within cancer cells. Its tankyrase inhibition disrupts the stability of AXIN, a scaffold protein that governs β-catenin degradation. By promoting β-catenin destruction, JPI-547 dampens oncogenic transcription pathways that fuel tumor growth and metastasis. This dual interference on both DNA repair and proliferative signaling pathways underlines the robust, multipronged mechanism of action, differentiating it decisively from the single-target approach of existing therapies.
Looking beyond efficacy, resistance mechanisms to conventional PARP inhibitors constitute a formidable clinical barrier. Tumors often develop secondary mutations restoring homologous recombination or activate compensatory pathways that negate inhibitor effects. JPI-547’s dual inhibitory mechanism offers a promising avenue to overcome such adaptive resistance by forcing tumor cells into a multifaceted molecular trap. Early preclinical evidence suggests delayed or reduced incidence of resistance development, though clinical validation remains a pressing imperative.
Another crucial aspect of JPI-547’s profile is the potential to act across multiple cancer types harboring BRCA mutations or exhibiting dysregulated Wnt signaling. While breast and ovarian cancers exemplify the initial targets, the compound’s dual mechanism could extend therapeutic benefits to malignancies such as pancreatic, prostate, and colorectal cancers, where similar molecular derangements drive pathogenesis. Such broad-spectrum applicability elevates the strategic value of JPI-547 in oncology pipelines.
The translational leap from preclinical promise to clinical reality will depend on rigorous trials assessing safety, tolerability, and efficacy in humans. Phase 1 clinical trials designed to establish dosing parameters and side effect profiles are eagerly anticipated. Beyond monotherapy, combination strategies involving immunotherapies, chemotherapeutics, or radiotherapy are plausible and could leverage synergistic effects, particularly given tankyrase’s role in modulating tumor immune microenvironments.
The holistic approach embodied by JPI-547 epitomizes a new era in personalized cancer treatments. By tailoring interventions to exploit unique genetic vulnerabilities and signaling dependencies within tumors, researchers move closer to precision oncology’s aspirational goals. The convergence of advanced medicinal chemistry, molecular biology, and cancer genetics within this compound’s development exemplifies the multidisciplinary innovation needed to tackle the world’s most formidable cancers.
Where once the horizon of cancer therapeutics seemed constrained by the limitations of single-target drugs, JPI-547 illuminates a future where sophisticated dual- and multi-target inhibitors become standard-bearers. These agents promise deeper, more durable responses that anticipate and forestall resistance, reshape tumor biology, and ultimately improve patient survival. The excitement surrounding JPI-547 is thus not merely about one drug, but about the broader shift it represents—a quantum leap in the design philosophy underlying cancer therapeutics.
As the scientific community continues to unravel the biological intricacies underscored by BRCA mutations and tumor signaling pathways like Wnt/β-catenin, the strategic targeting of multiple nodes within these systems will likely define next-generation therapies. JPI-547 stands at this intersection, a trailblazer in a class of compounds designed to outsmart cancer’s adaptive prowess. The potential clinical impact reinforces the importance of continued investment in innovative drug discovery and translational research.
In summary, the discovery and preclinical validation of JPI-547 offers an exciting, promising frontier in cancer treatment, particularly for BRCA1/2-mutated tumors that have historically eluded curative outcomes. Its dual inhibition of PARP1/2 and tankyrase not only heightens anti-cancer efficacy but also embodies a novel approach to overcoming resistance and broadening therapeutic scope. As the oncology field watches closely, JPI-547 may soon herald a new paradigm in the war against genetically complex malignancies, empowering clinicians and patients with more potent and durable weaponry.
Subject of Research: JPI-547 as a novel dual inhibitor of PARP1/2 and tankyrase in BRCA1/2-mutated cancer models.
Article Title: JPI-547, a novel dual inhibitor of PARP1/2 and tankyrase is more effective than first-generation PARP inhibitors in preclinical BRCA1/2-mutated cancer models.
Article References:
Kang, M.S., Katuwal, N.B., Ghosh, M. et al. JPI-547, a novel dual inhibitor of PARP1/2 and tankyrase is more effective than first-generation PARP inhibitors in preclinical BRCA1/2-mutated cancer models.
Br J Cancer (2026). https://doi.org/10.1038/s41416-026-03411-3
Image Credits: AI Generated
DOI: 06 May 2026
Tags: advancements in hereditary breast and ovarian cancer treatmentDNA repair inhibition in cancer treatmentdual-action inhibitors in cancer therapyJPI-547 dual PARP1/2 and tankyrase inhibitornext-generation PARnovel oncologic therapies for DNA damage repairovercoming resistance in PARP inhibitor therapysynthetic lethality in BRCA-deficient tumorstankyrase role in cancer progressiontargeted therapy for BRCA1/2-mutated cancersWnt/β-catenin signaling pathway targeting