Scientists are developing new materials that can both conduct electricity and repair heart muscle damaged during a heart attack
Credit: Michael Monaghan, Trinity College Dublin
WASHINGTON, D.C., October 8, 2019 — When a heart attack occurs, muscle in the heart tissue can be scarred, interfering with electrical activity necessary for healthy heart function. Drug treatments are available that alleviate further damage, but these don’t lead to tissue regeneration. Using artificial materials to patch or rebuild damaged parts has been tried but only recently has work focused on the electrical properties needed for proper cardiac operation.
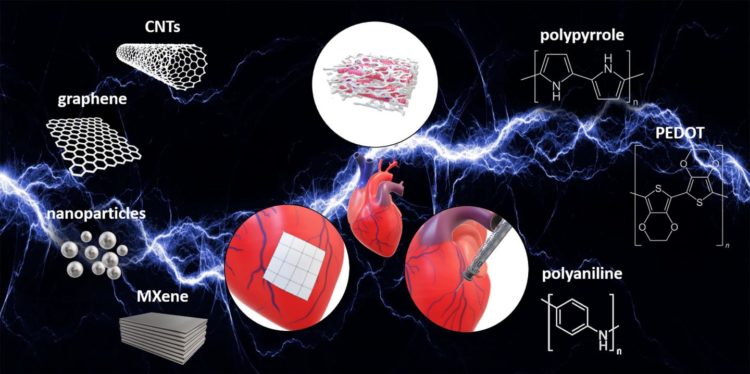
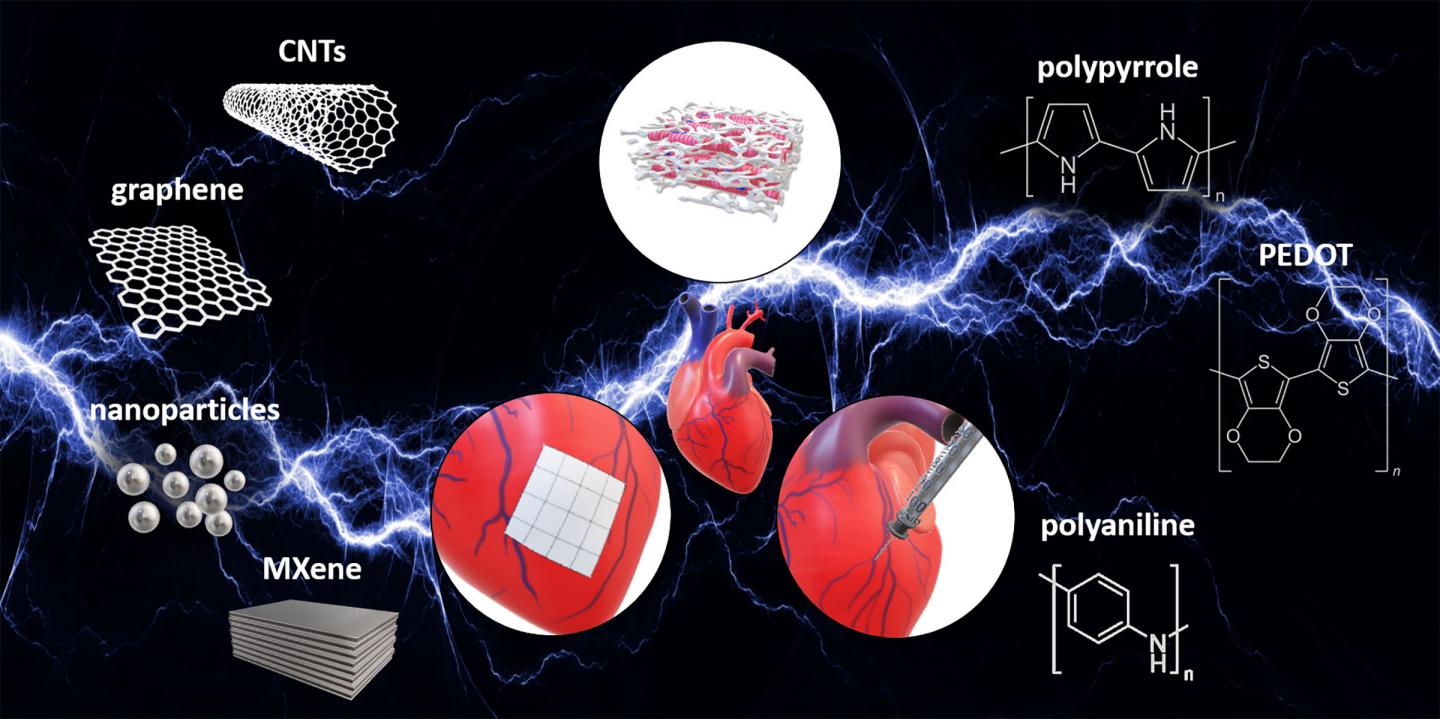
In this week’s APL Bioengineering, from AIP Publishing, investigators review the use of electrically conductive biomaterials for heart repair and treatment. The investigators considered three ways these materials can be used: to create scaffolds upon which heart cells might regenerate, to make electrically conductive patches to repair damaged tissue, and to produce injectable hydrogels to carry drugs to specific cardiac regions.
A healthy heart beats when cells in the myocardium contract. The myocardium is composed of precisely oriented fibers, so the contraction occurs in a twisting fashion. The contractions are triggered by an electrical signal from specialized cells known as the sinoatrial node. This signal is propagated through the heart muscle to the myocardium, unless it encounters scar tissue. The scar, which acts as an electrical insulator, may stop this signal, interfering with contraction.
Investigators are now developing electrically conductive materials to overcome this problem with scar tissue by matching the electrical conductance properties of native myocardium. The types of materials reviewed in this paper include small tubes or sheets of carbon; tiny metallic nanoparticles, usually of silver or gold; metal carbides, including the widely used titanium carbide; and polymers (plastics) doped with special substances that allow them to conduct electricity.
Any foreign material introduced into the body must be biocompatible to avoid both short- and long-term toxicity. Some toxicity issues are subtle. Silver nanoparticles, for example, have a size-dependent toxicity when delivered to the lungs. For many of these materials, toxicity, particularly long-term, has not yet been evaluated. However, some of these substances may impart beneficial effects. For example, certain metal carbides known as MXene may be anti-inflammatory.
Co-author Michael Monaghan suggests “a polymer known as PEDOT may be the most suitable for electrically conductive grafts or scaffolds, since it can be manufactured into 3D structures without different supporting materials.” The investigators also suggest some of PEDOT’s toxic properties might be prevented by more fully purifying the material when it is prepared. Further study is needed.
###
The article, “The rationale and emergence of electroconductive biomaterial scaffolds in cardiac tissue engineering,” is authored by Matteo Solazzo, Fergal J. O’Brien, Valeria Nicolosi and Michael G. Monaghan. The article will appear in the journal APL Bioengineering on Oct. 15, 2019 (DOI: 10.1063/1.5116579). After that date, it can be accessed at https:/
ABOUT THE JOURNAL
APL Bioengineering is an open access journal publishing significant discoveries specific to the understanding and advancement of physics and engineering of biological systems. See http://aip.
Media Contact
Larry Frum
[email protected]
301-209-3090
Related Journal Article
http://dx.