Pilot study of micro-fragmented adipose tissue injection suggests that the biologic is an effective treatment option for wheelchair users with chronic shoulder pain
Credit: Kessler Foundation/Jody Banks
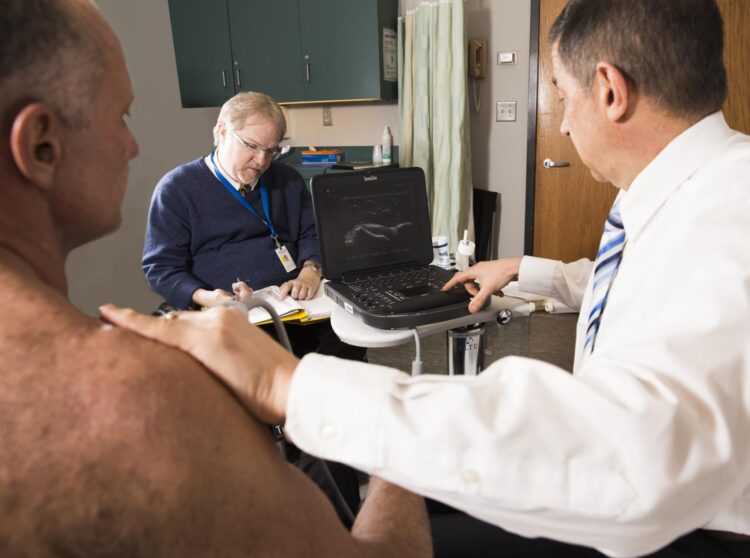
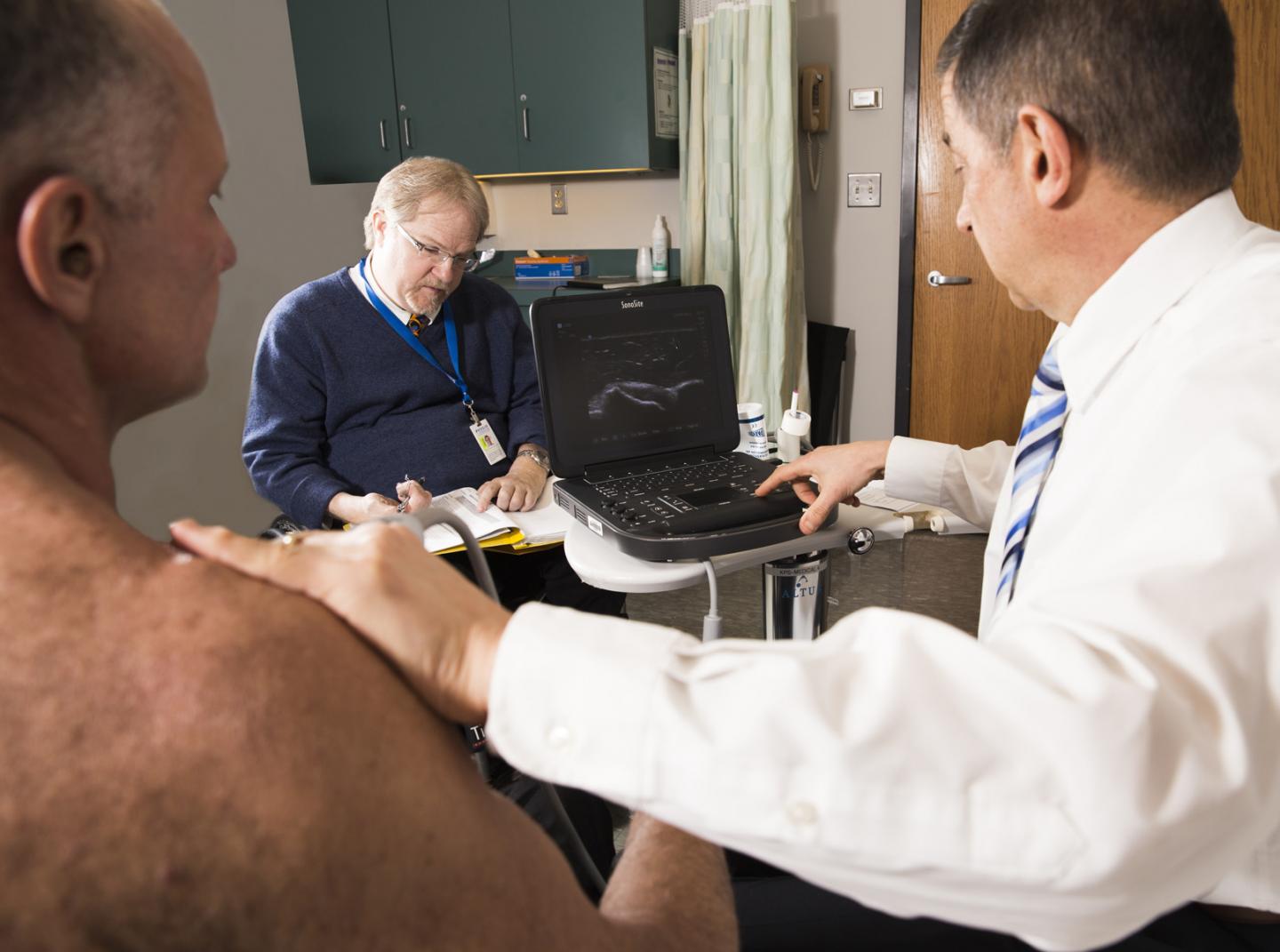
East Hanover, NJ. May 12, 2021. A team of specialists in regenerative rehabilitation conducted a successful pilot study investigating micro-fragmented adipose tissue (MFAT) injection for rotator cuff disease in wheelchair users with spinal cord injury. They demonstrated that MFAT injection has lasting pain-relief effects. The article, “A pilot study to evaluate micro-fragmented adipose tissue injection under ultrasound guidance for the treatment of refractory rotator cuff disease in wheelchair users with spinal cord injury,” (doi: 10.1080/10790268.2021.1903140) was published ahead of print on April 8, 2021, by the Journal of Spinal Cord Medicine.
The authors are Trevor Dyson-Hudson, MD, and Nathan Hogaboom, PhD, at Kessler Foundation; and Gerard Malanga, MD, a founder of the New Jersey Regenerative Institute and visiting scientist at Kessler Foundation, and Chris Cherian, MD, of Rutgers New Jersey Medical School. The study was conducted at the Derfner-Lieberman Laboratory for Regenerative Rehabilitation Research in the Center for Spinal Cord Injury Research at Kessler Foundation.
Shoulder pain is a common occurrence among wheelchair users with spinal cord injury because they rely solely on their upper limbs to perform everyday tasks. Often, pain is caused by soft-tissue injuries such as damage to rotator cuff tendons. Many non-surgical therapies for shoulder pain exist, including pain medication, physical therapy, and equipment modifications, but these have shown limited efficacy. Persistent shoulder pain can significantly lessen quality of life, and if conservative therapies fail, shoulder surgery is frequently the only option, which comes with its own set of risks and potential setbacks.
In this single-group pilot study, researchers explored the efficacy of a minimally invasive biological intervention involving an ultrasound-guided injection of MFAT, which harbors a potential source of bioactive and regenerative components for orthopedic conditions and may provide cushioning that can improve function and alleviate pain caused by rotator cuff injuries.
Ten wheelchair users with chronic spinal cord injury who had moderate-to-severe shoulder pain for more than six months caused by refractory rotator cuff disease participated in the study. All received an injection of MFAT and were evaluated at six and 12 months after treatment. Evaluation metrics included the 11-point Numerical Rating Scale, the Wheelchair User’s Shoulder Pain Index, Brief Pain Inventory pain interference items (BPI-17), Patient Global Impression of Change, ultrasound and physical examinations, and adverse events.
The results were encouraging, according to Drs. Hogaboom and Dyson-Hudson, co-directors of the Derfner-Lieberman Laboratory. Nearly 80 percent saw a meaningful decrease in pain symptoms, and all but one reported some improvement in pain and function. Moreover, scores declined steadily over the first three months for all metrics, and over the entire year for the BPI-17 metric, suggesting that this intervention has long-lasting effects. There were no significant adverse events.
“These results show that the minimally invasive injection of micro-fragmented adipose tissue is a safe and efficacious option for wheelchair users with shoulder pain caused by rotator cuff disease,” said Dr. Malanga. “Based on the success of our study, a randomized controlled study with a larger number of subjects has been initiated in this patient population through funding from the New Jersey Commission for Spinal Cord Research,” he added. ” We feel there is great potential for this therapy to help people with shoulder pain manage their symptoms and improve their quality of life. We credit our success to the Derfner Foundation for providing the initial funding to pursue this promising intervention, and acknowledge the ongoing efforts of the Alliance for Regenerative Rehabilitation Research & Training to advance the fields of rehabilitation sciences and regenerative medicine.”
###
Funding sources: Financial support was received in the form of a Pilot Grant and Fellowship Grant from the Derfner Foundation to conduct this pilot study. This research received funding from the Alliance for Regenerative Rehabilitation Research & Training (AR3T), which is supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), National Institute of Neurological Disorders and Stroke (NINDS), and National Institute of Biomedical Imaging and Bioengineering (NIBIB) of the National Institutes of Health under Award Number P2CHD086843.
About Kessler Foundation: Kessler Foundation, a major nonprofit organization in the field of disability, is a global leader in rehabilitation research that improves cognition, mobility and long-term outcomes, including employment, for people with neurological disabilities caused by diseases and injuries of the brain and spinal cord. Kessler Foundation leads the nation in funding innovative programs that expand opportunities for employment for people with disabilities. Learn more by visiting http://www.
Contact: Carolann Murphy, PA, . [email protected]
Media Contact
Carolann Murphy
[email protected]
Related Journal Article
http://dx.