A new compound with the potential to turn genes on and off could lead to new cancer and hereditary disease treatment strategies
Credit: Illustration by Mindy Takamiya (CC BY 4.0)
A new compound has the potential to bind to DNA and activate genes, which could lead to new treatments for cancers and hereditary diseases. Zutao Yu, Ganesh Pandian Namasivayam, and Hiroshi Sugiyama of Kyoto University’s Integrated Cell-Material Sciences (iCeMS) collaborated with colleagues in Japan and the USA to design and test a compound that could target specific DNA sequences and recruit gene-modifying molecules. Their findings were published in the journal ChemComm.
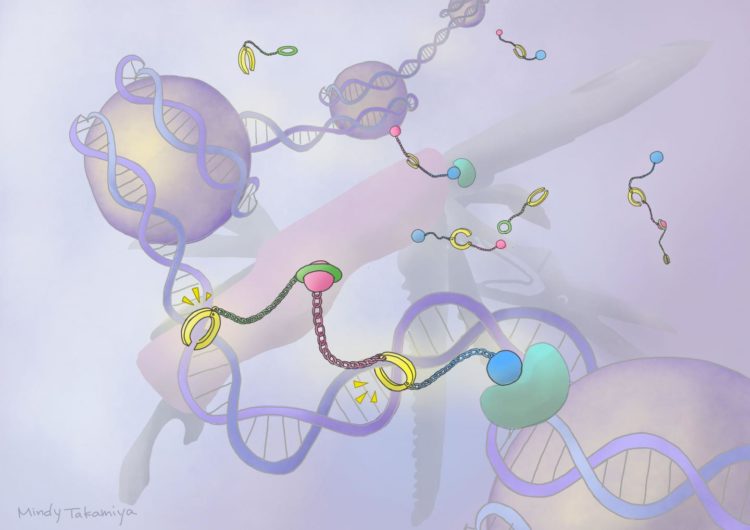
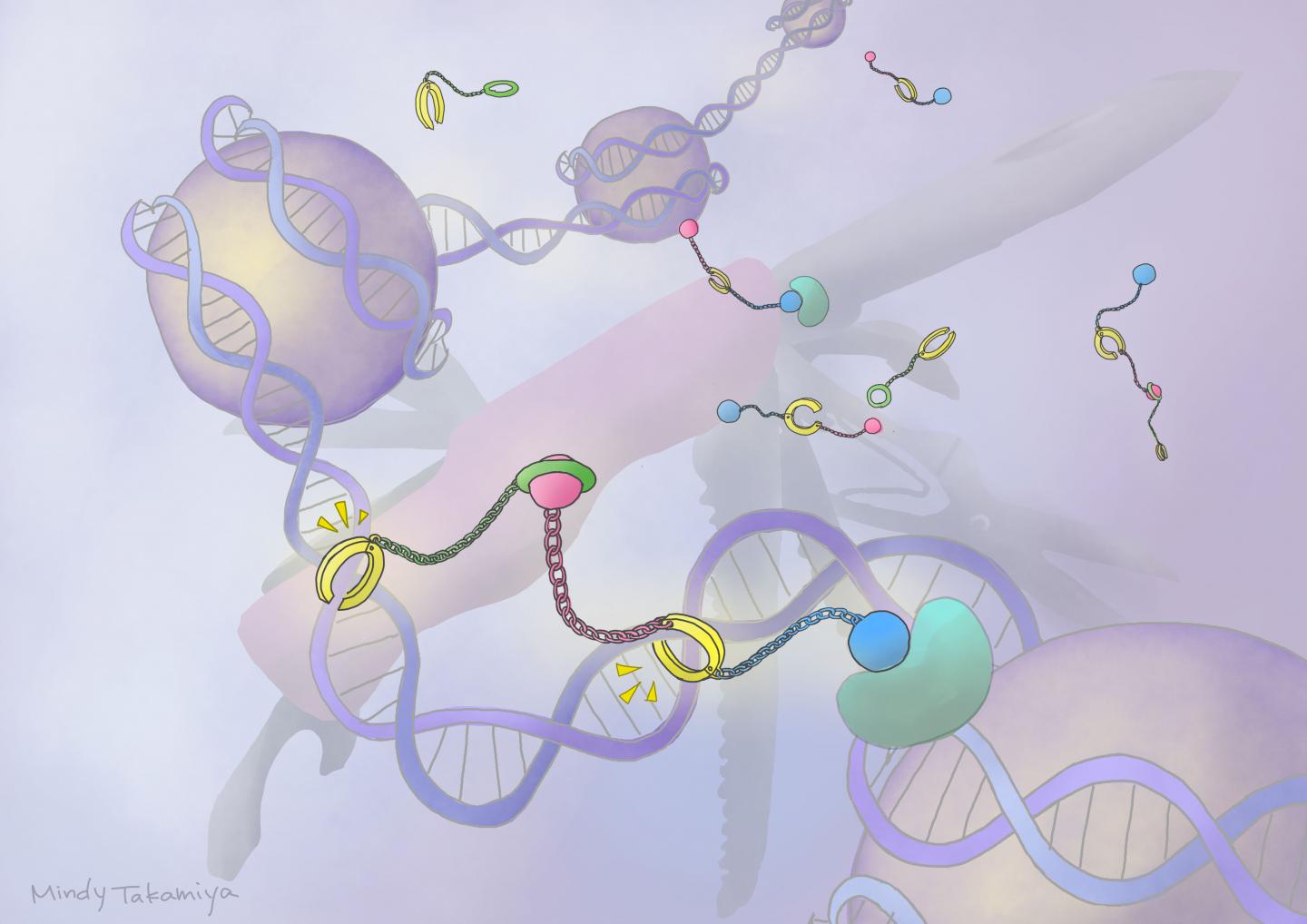
Scientists have long worked with small molecules called pyrrole-imidazole polyamides (PIPs) that have the ability to bind to minor grooves found in the DNA helix. They have been experimenting with PIPs as a drug-delivery mode that can switch genes on and off.
One such system involves combining a PIP with a ‘host-guest assembly’ (HoGu) that can strongly bind to DNA and act similarly to proteins, called transcription factors, that target and bind to specific genes to signal for them to turn on or off.
These PIP-HoGu systems can successfully mimic and disrupt transcription factor pairs from binding to DNA, resulting in a variety of biological effects. But they don’t have the ability to specifically activate genes.
Sugiyama and colleagues experimented with the idea of combining a PIP-HoGu system with an epigenetic drug, which can bind to and affect gene activation.
First, they improved the design of PIP-HoGu by selecting molecules that can strongly bind to DNA, while also being non-toxic, cell-permeable, water-soluble, and chemically stable. They then fine-tuned the molecules so that they targeted specific DNA nucleic acid sequences with flexible gap spacings.
The team next attached their new PIP-HoGu system to an epigenetic regulator molecule, forming what they refer to as ‘ePIP-HoGu’. They found that it more specifically bound to the targeted nucleic acid sequences and efficiently marked them for epigenetic modification.
“Our ePIP-HoGu is formed of three main components: a DNA-binding PIP, a cooperation domain, and an epigenetic modulator. Fine-tuning each of the three parts led to positive activity. Further optimization is required to explore its full potential and expand its biological and therapeutic applications,” says Yu.
This study builds on previous research to help advance PIPs as potential therapeutic drugs to address unmet needs in cancer treatment, rare hereditary diseases and in regenerative medicine, adds Namasivayam.
###
DOI: 10.1039/c9cc09608f
About Kyoto University’s Institute for Integrated Cell-Material Sciences (iCeMS)
At iCeMS, our mission is to explore the secrets of life by creating compounds to control cells, and further down the road to create life-inspired materials.
https:/
For more information, contact
I. Mindy Takamiya/ Mari Toyama
[email protected]
Media Contact
Mindy Takamiya
[email protected]
81-757-539-764
Related Journal Article
http://dx.