Various medical circumstances, including heart attacks and extreme cases of COVID-19, necessitate the use of anticoagulants, medicines that prevent blood clots. But the most commonly used, heparin, can induce potentially fatal side effects by making the blood clots worse rather than better. This only happens in a minority of patients so effective treatments are not commonly explored. For the first time, researchers, including those from the University of Tokyo, have proposed a side effect-free anticoagulating treatment that has so far proved effective in test mice and could be ready for human trials in just a few years.
Credit: ©2023 Yoshimoto et al. CC-BY
Various medical circumstances, including heart attacks and extreme cases of COVID-19, necessitate the use of anticoagulants, medicines that prevent blood clots. But the most commonly used, heparin, can induce potentially fatal side effects by making the blood clots worse rather than better. This only happens in a minority of patients so effective treatments are not commonly explored. For the first time, researchers, including those from the University of Tokyo, have proposed a side effect-free anticoagulating treatment that has so far proved effective in test mice and could be ready for human trials in just a few years.
The COVID-19 pandemic brought much woe to people around the world. And at the time of writing, though much of the world seems to have moved on, the effects of the pandemic continue to linger. One aspect of some extreme cases of COVID-19 that has not been widely reported is the complication brought on when the anticoagulant medicine heparin is used in an attempt to reduce blood clots in patients. A small number — up to 3% of recipients — suffer the side effect heparin-induced thrombocytopenia (HIT), a potentially fatal and rapid clotting of the blood, the opposite of the intended effect. Other medical issues, such as heart attacks, kidney dialysis, and even some surgeries, can also require anticoagulants.
Heparin was the first anticoagulant and is used widely — it’s considered incredibly important by the World Health Organization. But, because of the low number of HIT sufferers and thus lack of interest from the pharmaceutical industry, this issue is underexplored, despite its severity and increase due to COVID-19. It can be especially problematic in pregnant women as they cannot take existing treatments due to those potentially adversely affecting the fetus.
“The best treatment for HIT is an infusion of what are called thrombin inhibitors, but current drugs can lead to severe bleeding and there is no antidote to avoid this,” said Associate Professor Keitaro Yoshimoto from the Department of Life Sciences at the University of Tokyo. “Ideally, we could avoid HIT altogether. But at present, that is not possible, so we need a new low-risk thrombin inhibitor to replace current drugs. My team and I have created such an anticoagulant and have demonstrated it in mice and also in human blood plasma.”
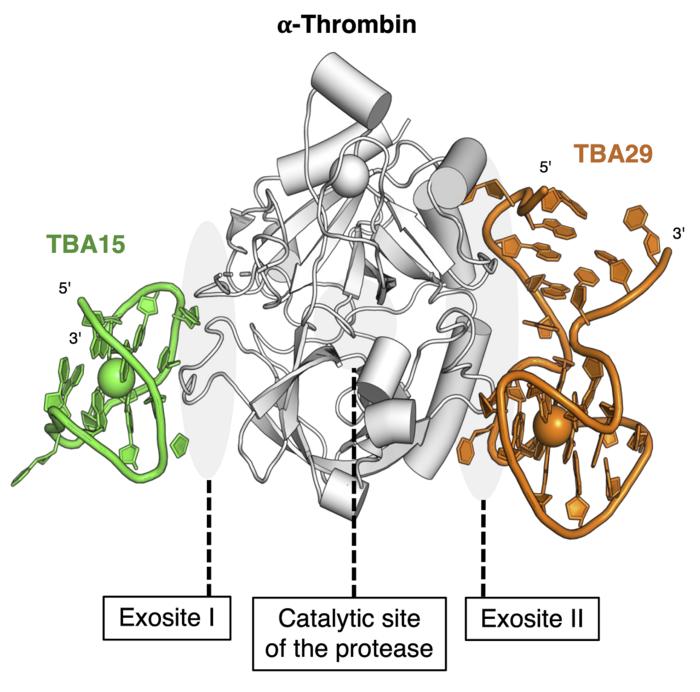
The team devised a next-generation thrombin inhibitor consisting of DNA molecules which includes a novel mechanism to prevent the severe bleeding. The key molecule in the drug is called a bispecific aptamer and its special feature is being able to bind to multiple things simultaneously. Another useful feature is short DNA sections which act as an antidote to the undesirable clotting side effect during HIT. This DNA-based drug essentially enables more complex behaviors than drugs based on simpler, more traditional chemistry. From their studies in mice, the team has shown the treatment is around 10 times as effective as the current best treatments for HIT. An additional benefit to pregnant women is that the nucleic acid-based drug and accompanying antidote do not cross the placenta to the fetus, as the DNA molecules in the drug are too large to cross the barrier presented by the placenta.
This research came about as Yoshimoto has a history in biochemistry and the science of molecular separation, specializing in a method called MACE®-SELEX for the selection of aptamers, short sections of DNA useful for medicine. He teamed up with Assistant Professor Asuka Sakata of Nara Medical University in Japan who specializes in thrombosis biology, and together with their team, they embarked on using Yoshimoto’s ideas to solve medical issues raised in Sakata’s research.
“We hope to proceed with human trials soon,” said Yoshimoto. “It will take up to two years for preclinical studies and five years to complete clinical trials in humans. Though the number of HIT sufferers is small, it’s such a severe condition I feel it’s important we tackle it quickly.”
###
Journal article: Masanobu Nagano, Kazuki Kubota, Asuka Sakata, Rei Nakamura, Toru Yoshitomi, Koji Wakui, Keitaro Yoshimoto. “A neutralizable dimeric anti-thrombin aptamer with potent anticoagulant activity in mice”, Molecular Therapy – Nucleic Acids, https://doi.org/10.1016/j.omtn.2023.07.038
Funding:
K.Y. and A.S. received grants from AMED under Grant Number JP22ak0101130. K.Y. received grants from the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (B) (18H02002) and Grant-in-Aid for Transformative Research Areas (22H05049), and SENSHIN Medical Research Foundation. M.N. received grants from JSPS Grant-in-Aid for Scientific Research (C) (21K06450), Iketani Science and Technology Foundation, Foundation for Interaction in Science & Technology, and The Research Foundation for Pharmaceutical Sciences.
Declaration of interests:
M.N., K.K., A.S., and K.Y. are involved in patent application related to this study (JP2022/212461). A.S.: members of Medicinal Biology of Thrombosis and Hemostasis established by Nara Medical University and Chugai Pharmaceutical Co., Ltd.; speaker’s bureau from CSL Behring. K.Y. is the CTO of LinkBIO Co., Ltd. K.Y. collaboratively works under The University of Tokyo-LinkBIO Co., Ltd. or The University of Tokyo-Nara Medical University-Chugai Pharmaceutical Co., Ltd. or LinkBIO Co., Ltd.- Nara Medical University-Chugai Pharmaceutical Co., Ltd. The other authors declare no competing financial interest.
Departmental links:
Graduate School of Arts and Sciences – https://www.c.u-tokyo.ac.jp/eng_site/
Department of Life Sciences – https://bio.c.u-tokyo.ac.jp/english.html
Research contact:
Associate Professor Keitaro Yoshimoto
Department of Life Sciences, Graduate School of Arts and Sciences,
The University of Tokyo, 3-8-1 Komaba, Meguro-ku, Tokyo 153-8902, Japan
[email protected]
Press contact:
Mr. Rohan Mehra
Public Relations Group, The University of Tokyo,
7-3-1 Hongo, Bunkyo-ku, Tokyo 113-8656, Japan
[email protected]
About The University of Tokyo
The University of Tokyo is Japan’s leading university and one of the world’s top research universities. The vast research output of some 6,000 researchers is published in the world’s top journals across the arts and sciences. Our vibrant student body of around 15,000 undergraduate and 15,000 graduate students includes over 4,000 international students. Find out more at www.u-tokyo.ac.jp/en/ or follow us on Twitter at @UTokyo_News_en.
Journal
Molecular Therapy
DOI
10.1016/j.omtn.2023.07.038
Method of Research
Experimental study
Subject of Research
Human tissue samples
Article Title
A neutralizable dimeric anti-thrombin aptamer with potent anticoagulant activity in mice
Article Publication Date
1-Aug-2023
COI Statement
M.N., K.K., A.S., and K.Y. are involved in patent application related to this study (JP2022/212461). A.S.: members of Medicinal Biology of Thrombosis and Hemostasis established by Nara Medical University and Chugai Pharmaceutical Co., Ltd.; speaker’s bureau from CSL Behring. K.Y. is the CTO of LinkBIO Co., Ltd. K.Y. collaboratively works under The University of Tokyo-LinkBIO Co., Ltd. or The University of Tokyo-Nara Medical University-Chugai Pharmaceutical Co., Ltd. or LinkBIO Co., Ltd.- Nara Medical University-Chugai Pharmaceutical Co., Ltd. The other authors declare no competing financial interest.




