A novel experimental drug known as DH20931 has emerged as a promising candidate in the battle against triple-negative breast cancer (TNBC), one of the most formidable and aggressive forms of breast cancer due to its lack of common therapeutic targets. Recent preclinical investigations conducted by an international team led by Dr. Satya Narayan at the University of Florida have demonstrated the drug’s unique mechanism of overwhelming malignant cells with an onslaught of toxic lipid molecules, specifically ceramides, thereby triggering a cascade of cellular stress and programmed cell death.
TNBC is notoriously difficult to treat because it does not express estrogen receptors, progesterone receptors, or HER2, making it unresponsive to hormone therapies or HER2-targeted agents that have revolutionized treatment for other breast cancer subtypes. Consequently, chemotherapy remains the mainstay of treatment, although often with limited efficacy and significant side effects. The advent of DH20931 introduces a novel therapeutic angle — metabolic disruption via lipid induction — which may tilt the balance in favor of eradicating cancer cells while sparing normal tissue.
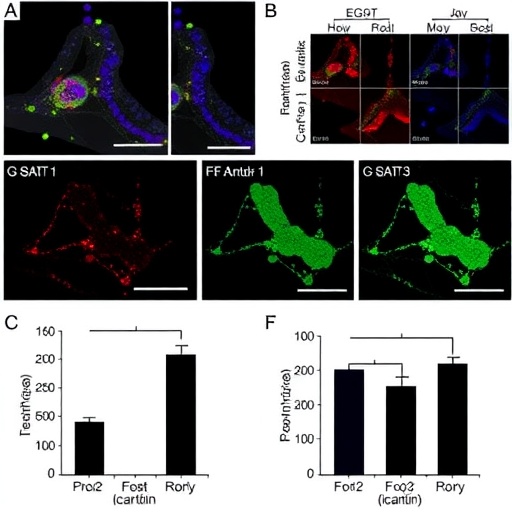
At the heart of DH20931’s mechanistic action lies its capability to target Ceramide Synthase 2 (CerS2), an enzyme pivotal in the synthesis of ceramides, a class of sphingolipids that regulate numerous cellular functions including apoptosis, proliferation, and differentiation. By pharmacologically activating CerS2, DH20931 causes an accumulation of ceramide molecules within the lipid bilayer of cancer cells, essentially flooding them with molecular “fats” that induce significant cytotoxic stress. This lipid overload disrupts membrane integrity and intracellular signaling, pushing already stressed cancer cells beyond their metabolic limits.
The research, recently published in Molecular Cancer Therapeutics, details how human-derived TNBC tumors implanted into murine models showed markedly diminished growth when treated with DH20931. Notably, the drug achieved this without causing overt toxicity or weight loss in animal subjects, an encouraging sign that selective cytotoxicity might be attainable. Furthermore, DH20931 exhibited enhanced anticancer activity when combined with conventional chemotherapy drug doxorubicin, allowing for a fivefold reduction in the required chemotherapy dose to achieve effective tumor cell killing.
Beyond ceramide accumulation, DH20931 exerts a secondary cytotoxic mechanism through the modulation of intracellular calcium levels. The drug induces a calcium surge within cancer cells, which disrupts mitochondrial homeostasis – the critical hub of energy production and apoptosis regulation. By compromising mitochondria simultaneously through lipid and calcium stress, DH20931 initiates a synergistic “two-hit” cytotoxic effect, ensuring that the malignant cells face an insurmountable metabolic crisis leading to their demise.
This dual-pathway mode of action exploits fundamental vulnerabilities in cancer cell metabolism and stress response. Dr. Narayan likens the process to an electrical system overwhelmed by a power surge: while healthy cells behave like well-grounded circuits with protective fuses, cancer cells are akin to chaotic wiring prone to short-circuiting under excess strain. In this analogy, DH20931 delivers a flood of toxic lipids and calcium, analogous to a surge of electricity, which “burns out” the cancer cells’ protective mechanisms, leading to their self-destruction.
Significantly, the research team observed that normal cells exhibit comparatively lower sensitivity to DH20931, underscoring the therapeutic window that makes this compound particularly attractive. The selective targeting may stem from cancer cells’ already elevated basal stress and altered lipid metabolism, which render them less capable of handling additional biochemical insults compared to their healthy counterparts.
The origins of DH20931 trace back to a synthetic chemistry lab led by Dr. Sukwong Hong at the Gwangju Institute of Science and Technology in South Korea. Dr. Hong’s work in generating novel CerS2-targeting molecules paved the way for collaborations with Dr. Narayan’s group, who conducted rigorous biological evaluations for efficacy and safety. The interdisciplinary effort harnessed medicinal chemistry, cell biology, and tumor modeling to validate DH20931’s potential.
Although the current findings are compelling, extensive preclinical validation and ultimately clinical trials in humans are essential to ascertain DH20931’s safety, pharmacokinetics, and therapeutic efficacy in patients. Triple-negative breast cancer presents unique challenges, including heterogeneity and propensity for metastasis, which necessitate a multi-pronged treatment approach. The ability of DH20931 to enhance chemotherapy response also suggests it could become a valuable adjunct, potentially lowering chemotherapy-associated toxicities.
The implications of this research extend beyond TNBC. Given that the metabolic and mitochondrial pathways targeted by DH20931 are implicated across various solid tumors, this drug could form the basis of a new class of cancer therapeutics that leverage metabolic vulnerabilities through lipid and calcium dysregulation. The researchers aim to explore the drug’s efficacy in other breast cancer subtypes and diverse malignancies, broadening its therapeutic horizon.
This innovative approach, combining molecular targeting of ceramide production with mitochondrial disruption, heralds a paradigm shift in oncologic drug development. The study underscores the critical importance of metabolic stress pathways in cancer survival and opens avenues for exploiting these pathways to achieve selective tumor cell killing with minimal harm to normal tissues.
In sum, DH20931 represents a beacon of hope in the treatment landscape of triple-negative breast cancer, an area that historically has lacked targeted therapies and suffered from poor patient prognosis. By exploiting the cancer cells’ metabolic fragility through a sophisticated two-hit mechanism, this drug exemplifies the kind of ingenious therapeutic strategies needed to conquer aggressive cancers.
The research was published April 21, 2026, in Molecular Cancer Therapeutics, and the findings were disseminated at the American Association for Cancer Research’s annual meeting in San Diego, highlighting the broader scientific community’s interest in this exciting development.
Subject of Research: Animals
Article Title: CerS2 is a druggable target in triple-negative breast cancer
News Publication Date: 21-Apr-2026
Web References: http://dx.doi.org/10.1158/1535-7163.MCT-25-1159
References: Narayan, S. et al. CerS2 is a druggable target in triple-negative breast cancer. Molecular Cancer Therapeutics (2026).
Keywords: Triple-negative breast cancer, CerS2, Ceramides, DH20931, Drug development, Chemotherapy enhancement, Metabolic targeting, Mitochondrial stress
Tags: Ceramide Synthase 2 targetingceramide-induced cancer cell deathchallenges in treating triple-negative breast cancerDH20931 drug mechanismexperimental breast cancer drugmetabolic disruption in cancer therapynovel TNBC therapeutic approachespreclinical breast cancer drug studiesprogrammed cell death by lipidssphingolipid metabolism in cancertoxic lipid therapy for cancertriple negative breast cancer treatment