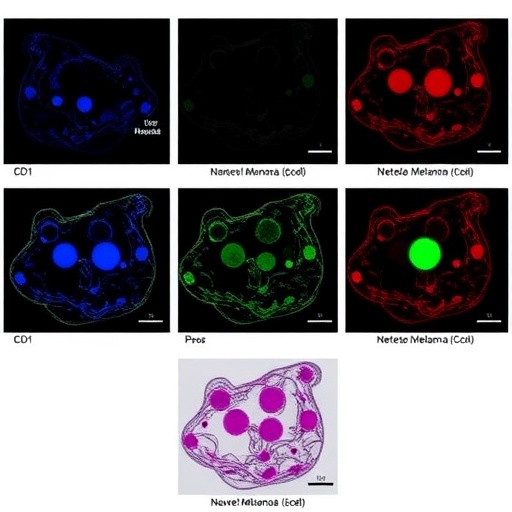
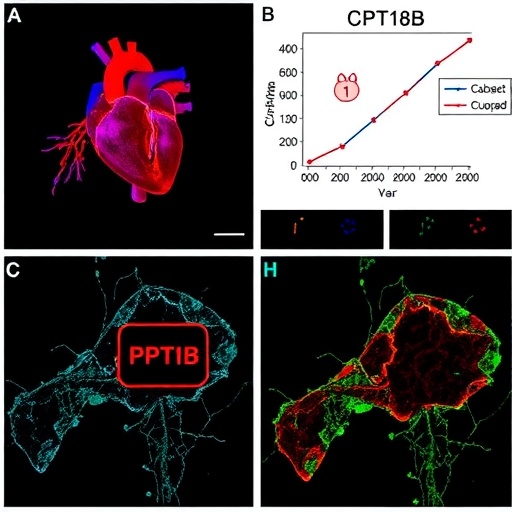
A groundbreaking study published in the renowned journal Oncotarget reveals compelling evidence that the inhibition of stearoyl-CoA desaturase-1 (SCD1) by aramchol synergizes with the multi-kinase inhibitor regorafenib and the metabolic drug metformin to robustly enhance tumor cell death. This intriguing intersection of targeted therapies unveils a novel avenue for tackling challenging malignancies such as uveal melanoma (UM) and cholangiocarcinoma, notorious for their resistance to conventional treatments. Led by scientists Michael R. Booth, Laurence Booth, and Jane L. Roberts at Virginia Commonwealth University, with collaborative input from the University of Pittsburgh Cancer Institute, the study highlights a multifaceted cell death mechanism that integrates macroautophagy and apoptotic signaling pathways.
At the heart of this research lies the SCD1 inhibitor aramchol, a compound known for its ability to modulate lipid metabolism by suppressing the enzyme responsible for the desaturation of saturated fatty acids into monounsaturated fatty acids, an essential step in membrane biosynthesis and signaling. Aramchol’s interaction with regorafenib and metformin was systematically investigated in patient-derived UM cells and LD-1 cholangiocarcinoma models, representing clinically relevant tumor types. The authors demonstrated that combining these agents significantly intensified tumor cell mortality beyond the efficacy of each compound used individually.
The mechanistic underpinnings of this enhanced cytotoxic effect were traced to an upregulation of autophagic flux and autophagosome formation. Autophagy, a cellular catabolic process typically serving homeostatic functions, here contributes to a lethal response when excessively activated or dysregulated. Experimental knockdown of key autophagy-related proteins, including Beclin1, ATG5, and LAMP2, markedly diminished autophagosome formation and lowered tumor cell death rates, therefore establishing the indispensability of macroautophagy in mediating the observed therapeutic synergy.
Intriguingly, the pro-apoptotic BH3-interacting domain death agonist (BID) emerged as an essential component of the cytotoxic interplay. BID is known for its role in death receptor-mediated apoptosis and mitochondrial outer membrane permeabilization, suggesting that the therapeutic combination activates programmed cell death through a convergence of autophagy and classical apoptotic signals. Silencing BID expression attenuated cell death induced by the drug combination, indicating its pivotal role as a signaling node bridging autophagic processes and apoptosis.
Beyond autophagy and apoptosis, the study explored the distinct molecular intricacies engendered by aramchol. Although SCD1 knockdown alone elevated baseline tumor cell death, it failed to recapitulate the complete anticancer activity elicited by aramchol, implying that this agent must engage additional, yet unidentified, molecular targets. Such polypharmacology may underlie aramchol’s efficacy and warrants further molecular characterization.
One of the study’s most striking revelations is the pronounced enhancement of tumor cell killing when metformin is added to the aramchol and regorafenib combination. Metformin, widely recognized as an antidiabetic drug, has garnered interest for its potential anticancer properties linked to modulation of mitochondrial metabolism and AMP-activated protein kinase (AMPK) signaling. In this context, metformin seemingly augments autophagic flux, amplifying the cytotoxic cascading events initiated by aramchol and regorafenib.
Detailed viability assays employing trypan blue exclusion confirmed that the triple-drug regimen dramatically reduced cell survival in multiple independent experiments involving UM and HEP3B hepatocellular carcinoma cell lines. These findings underscore the robustness and reproducibility of the therapeutic effect, lending significant translational promise to this approach.
Furthermore, the research team emphasized the criticality of macroautophagy as a double-edged sword in cancer biology. Whereas basal autophagy often supports tumor survival, excessive induction can trigger autophagic cell death or sensitize cells to apoptosis, strategically exploited here by the pharmacologic combination. This nuanced manipulation of autophagy pathways could be pivotal in overcoming resistance mechanisms endemic to metastatic UM, a malignancy with notoriously poor prognosis once disseminated to the liver.
Recognizing its potential clinical implications, the team advocates for in vivo assessment of this combinatorial therapy. Particularly in metastatic UM, where effective treatments remain elusive, exploiting synergistic drugs capable of modulating both metabolic and signaling networks might enable more substantial tumor control, especially within the liver microenvironment, which frequently harbors metastatic deposits.
Importantly, the research also addresses safety and translational feasibility by underscoring the specific molecular targets involved and the necessity of maintaining a balance between efficacy and normal tissue toxicity. As aramchol is currently under clinical evaluation for other indications, its repurposing in oncology, in combination with clinically approved agents such as regorafenib and metformin, could expedite therapeutic development.
The study’s comprehensive mechanistic insights and promising preclinical results set a new standard for targeted combination therapy in oncology. This multifactorial approach, leveraging macroautophagy and death receptor signaling, opens a frontier for precision medicine strategies tailored to recalcitrant tumors like uveal melanoma and cholangiocarcinoma, ultimately aiming to translate benchside discoveries into lifesaving clinical outcomes.
For those interested in delving into the full scientific details, the original research article is accessible via the Digital Object Identifier (DOI) link: https://doi.org/10.18632/oncotarget.28861. Correspondence and inquiries about this transformative study can be directed to the lead author, Dr. Paul Dent ([email protected]), at Virginia Commonwealth University.
Subject of Research:
Article Title: The SCD1 inhibitor aramchol interacts with regorafenib and metformin to kill tumor cells
News Publication Date: 27-Mar-2026
Web References: https://doi.org/10.18632/oncotarget.28861
Image Credits: Copyright © 2026 Booth et al. Courtesy of Virginia Commonwealth University and Galmed Pharmaceuticals Ltd.
Keywords: cancer, macroautophagy, ER stress, aramchol, regorafenib, BID
Tags: cholangiocarcinoma combined treatmentcombined therapy uveal melanomalipid metabolism in cancer cellsmacroautophagy and apoptosis in tumor cellsmetabolic drugs enhancing cancer cell deathmulti-kinase inhibitors in cancer therapynovel cancer cell death mechanismsovercoming drug resistance in melanomapatient-derived tumor cell modelsregorafenib and metformin synergySCD1 inhibitor aramchol cancer treatmentuveal melanoma targeted therapies