In the intricate network of the human biliary system, the accurate diagnosis of bile duct strictures remains a formidable challenge for clinicians. These narrowings or blockages, connecting vital organs such as the liver, gallbladder, and intestines, can herald either malignant or benign processes. Determining the precise etiology is crucial, as it directs therapeutic strategies ranging from surgical intervention to careful monitoring. Traditional diagnostic modalities like biopsy and cytology, while standard, often fall short—particularly due to the difficult anatomical location and the presence of inflammation or fibrosis masking underlying malignancies. This diagnostic uncertainty can lead to delayed treatment and increased patient morbidity.
Addressing this critical gap, researchers at the UPMC Hillman Cancer Center and the University of Pittsburgh School of Medicine have pioneered an innovative molecular diagnostic tool known as BiliSeq. This next-generation sequencing (NGS) test leverages genetic mutation detection to improve early and accurate identification of bile duct cancers. Published in the prestigious journal Gastroenterology, the study evaluates BiliSeq’s real-world clinical performance over six years, involving a large cohort of over 2,000 patients and nearly 3,000 bile duct specimens sourced from across the United States.
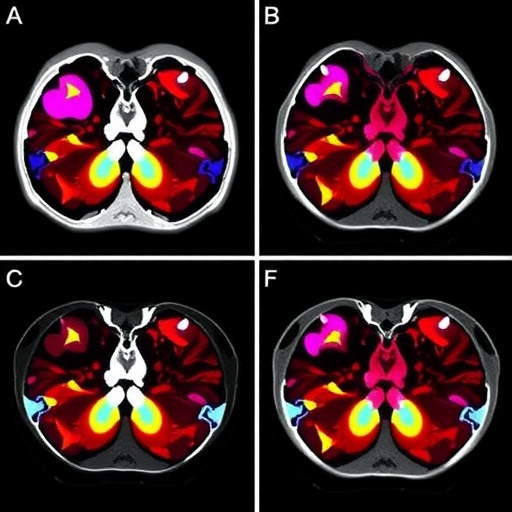
BiliSeq operates by analyzing both DNA and RNA sequences extracted from bile duct tissue samples to detect neoplastic genetic alterations. Tumors in the bile ducts are notoriously scarce in cellularity and can be obscured by reactive changes. Consequently, conventional microscopic techniques frequently produce false-negative results. The molecular approach incorporated in BiliSeq transcends this limitation by identifying oncogenic mutations even in the absence of abundant malignant cells. As a result, the test demonstrated approximately double the sensitivity compared to pathology alone, detecting about 82% of bile duct malignancies in the studied sample group.
Importantly, when BiliSeq results were integrated with traditional pathological assessment, cancer detection rates increased further to nearly 90%. This synergistic diagnostic approach not only enabled clinicians to confidently exclude cancer when absent but also minimized the risk of misclassifying benign inflammatory conditions as cancer, thereby preventing unnecessary invasive procedures. These findings signify a transformative shift toward enhanced precision in the evaluation of biliary strictures.
One of the remarkable features of BiliSeq is its capacity to deliver detailed molecular profiles that go beyond a binary cancer diagnosis. The study revealed that approximately 20% of patients had actionable genetic alterations identified by BiliSeq, offering the potential for personalized therapeutic interventions. In about one third of these cases, clinicians adapted treatment plans based on the genetic insights, underscoring BiliSeq’s significant role in advancing personalized medicine within hepatobiliary oncology.
This genomic approach also holds substantial promise for high-risk populations, including patients with primary sclerosing cholangitis (PSC) and individuals of Hispanic descent, groups that historically experience lower detection rates with standard pathology. In these subpopulations, reliance on pathology alone could miss up to half of the cancer cases. However, incorporation of BiliSeq bolstered diagnostic sensitivity to as high as 86%, markedly improving early cancer identification and subsequent patient outcomes.
The development and validation of BiliSeq emerge from a greater movement within academic medicine to harness molecular diagnostics for complex cancers where anatomical and biological obstacles impede conventional evaluation. Previous work by Pitt and UPMC researchers has advanced molecular tools for pancreatic malignancies and precancerous cystic lesions, spearheaded by co-author Aatur Singhi, M.D., Ph.D. The latest findings not only cement BiliSeq as a robust diagnostic adjunct but also highlight the collaborative, multidisciplinary efforts essential for innovation in cancer diagnostics.
Methodologically, the study distinguishes itself with its prospective, multi-institutional, and large-scale nature, providing robust evidence that reflects routine clinical practice rather than isolated laboratory conditions. The authors emphasize that samples analyzed originate from diverse medical centers nationwide, enhancing the generalizability and clinical applicability of the results. This real-world framework is critical for ensuring that the test’s benefits translate into tangible improvements in patient care.
Furthermore, BiliSeq findings are already influencing complex clinical decisions, such as liver transplantation candidacy for select patients evaluated at UPMC. Here, molecular data helping to characterize tumor biology and cancer risk may optimize transplant outcomes, reducing both recurrence rates and unwarranted exclusion due to diagnostic uncertainty. This integration of precision medicine tools exemplifies the trajectory of modern oncology toward individualized care pathways.
While BiliSeq is not intended as a universal screening modality for the general population, its utility for patients presenting with bile duct strictures is profound. By reducing diagnostic ambiguity, the test minimizes the need for repetitive invasive sampling procedures, decreases waiting times for definitive diagnosis, and broadens therapeutic options. Each of these factors contributes not only to better clinical outcomes but also to less patient anxiety and healthcare resource optimization.
The study’s extensive author team, which includes experts in gastroenterology, pathology, oncology, and surgery from both UPMC and the University of Pittsburgh, reflects the interdisciplinary nature essential to advancing cancer diagnosis and management. Funded by prominent institutions including the National Institutes of Health and foundational cancer organizations, this research exemplifies a model of translational science steadily moving innovations from bench to bedside.
As molecular diagnostics continue to revolutionize cancer care, BiliSeq stands out as a pioneering example of next-generation sequencing technology applied to a challenging and rare form of cancer. Its ability to uncover subtle genetic malignancies, guide personalized treatment, and support complex clinical decision-making makes it a beacon of hope for patients facing the uncertainty of bile duct strictures. With continued refinement and adoption, this technology may soon become the new standard, fundamentally changing the diagnostic landscape in hepatobiliary oncology.
Subject of Research: Molecular diagnostics in bile duct cancer
Article Title: DNA/RNA-Based Next-Generation Sequencing (NGS) Improves the Early Diagnosis and Management of Neoplastic Bile Duct Strictures: A Six-Year, Prospective, Multi-Institutional, Real-Time Study
News Publication Date: 27-Mar-2026
Web References: www.upmc.com/media
References: Published in Gastroenterology, DOI: 10.1053/j.gastro.2026.02.040
Image Credits: Credit UPMC and University of Pittsburgh Health Sciences
Tags: advanced molecular testing in gastroenterologybile duct biopsy limitationsbile duct cancer detection methodsbile duct cancer diagnostic advancementsbile duct cancer early detection methodsbile duct stricture diagnosisbile duct stricture diagnosis challengesbile duct tissue sequencingBiliSeq molecular testchallenges in bile duct cancer diagnosisclinical performance of BiliSeq testearly detection of bile duct tumorsgenetic mutation analysis in bile duct cancergenetic mutation detection in biliary cancerimproving bile duct cancer diagnosis accuracyimproving sensitivity of cancer detection testsmolecular diagnostic test for bile duct cancermolecular diagnostic tools for bile duct cancermulti-institutional study on bile duct cancernext-generation sequencing in cancer detectionnext-generation sequencing in cancer diagnosisUPMC Hillman Cancer Center bile duct researchUPMC Hillman Cancer Center research