A groundbreaking beacon in pancreatic cancer research is shining brighter with the emergence of glutamic-oxaloacetic transaminase 2 (GOT2) as a pivotal therapeutic target. Pancreatic cancer notoriously ranks among the deadliest and most therapeutically unyielding malignancies, evading conventional treatments with alarming efficacy. At the core of this resilience lies the unique metabolic circuitry that cancer cells exploit, and GOT2 has come into sharp focus as a mitochondrial enzyme whose multifaceted roles are critical to tumor viability and immune evasion.
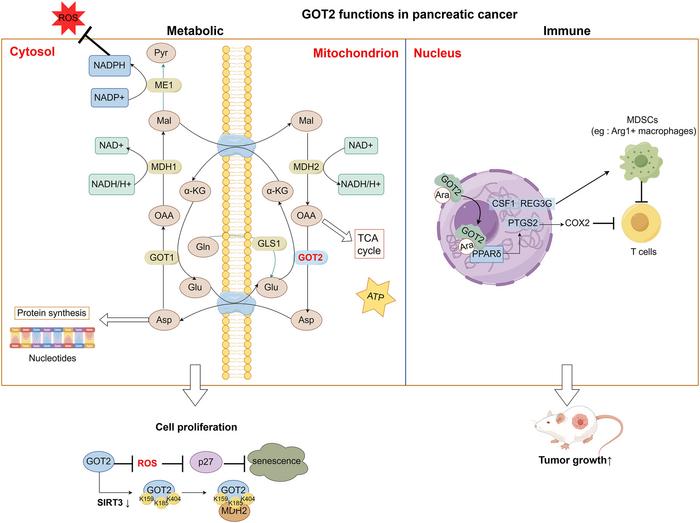
GOT2 functions primarily within the mitochondrial matrix as a key enzyme in the transamination of glutamate and oxaloacetate, facilitating the malate-aspartate shuttle — a fundamental pathway for maintaining the cellular NAD+/NADH redox balance. This enzymatic activity supports the continuous regeneration of NAD+, which is indispensable for glycolysis and mitochondrial oxidative phosphorylation, effectively sustaining the energetic and anabolic demands of proliferating pancreatic cancer cells. Crucially, GOT2’s metabolic products, particularly aspartate and α-ketoglutarate, feed directly into nucleotide biosynthesis and the tricarboxylic acid (TCA) cycle, underscoring its centrality in promoting biosynthetic and bioenergetic homeostasis.
Unlike many cancers that predominantly utilize canonical glutamine metabolism, pancreatic tumors harboring oncogenic KRAS mutations display a distinctive reliance on GOT2-mediated metabolic reprogramming. This non-canonical glutamine metabolism circumvents traditional glutamate dehydrogenase pathways, instead harnessing GOT2 activity to sustain intracellular pools of aspartate and other intermediates, which become bottlenecks in anabolic processes and cell cycle progression. Consequently, inhibiting GOT2 not only halts these biosynthetic pathways but also disrupts mitochondrial redox buffering, leading to reactive oxygen species (ROS) accumulation, oxidative stress, and ultimately cellular senescence or apoptosis.
Intriguingly, recent studies have uncovered an unanticipated nuclear function of GOT2 that transcends its canonical metabolic role. In the nucleus, GOT2 has been identified as a fatty acid transporter that activates peroxisome proliferator-activated receptor delta (PPARδ), a nuclear transcription factor implicated in lipid metabolism, inflammation, and immune modulation. This activity initiates a transcriptional program upregulating immunosuppressive genes including PTGS2 (encoding COX-2), CSF1, and REG3G, thereby sculpting a tumor microenvironment hostile to effective T-cell infiltration and anti-tumor immunity. GOT2’s dual capacity as both metabolic enzyme and immune modulator establishes it as a nexus connecting metabolic and immune evasion mechanisms.
Despite this promising targetability, pancreatic tumors reveal a formidable capacity for adaptive resistance to GOT2 inhibition. Tumor cells exploit macropinocytosis — a form of bulk nutrient uptake — to salvage extracellular metabolites, particularly aspartate, circumventing the metabolic blockade imposed by GOT2 loss. Additionally, metabolic crosstalk with cancer-associated fibroblasts (CAFs) supplies alternative sources of essential metabolites, enabling tumor survival and continued proliferation even under targeted metabolic stress. These findings underscore the complexity of pancreatic tumor metabolism and the necessity for combinatorial therapeutic approaches.
Progress in the development of potent and selective GOT2 inhibitors is accelerating, with compounds such as amino oxyacetate emerging as early candidates demonstrating efficacy in preclinical models. Such inhibitors represent an enticing avenue to cripple the metabolic flexibility of pancreatic cancer cells. Moreover, integrating GOT2-targeted therapies with immune checkpoint inhibitors or agents modulating oxidative stress could potentiate therapeutic responses by simultaneously dismantling metabolic support and immunosuppressive barriers within tumors.
The structural biology of GOT2 provides critical insights that facilitate rational drug design. As a pyridoxal phosphate-dependent aminotransferase, GOT2’s active site architecture and substrate binding dynamics offer multiple intervention points. Targeting enzyme cofactor interactions or substrate analog competition may yield inhibitors with enhanced specificity and reduced off-target effects, pivotal for clinical translation. Advances in high-resolution crystallography and computational modeling continue to illuminate these possibilities, accelerating medicinal chemistry efforts.
Furthermore, the metabolic plasticity exhibited by pancreatic cancer cells highlights the intricate interplay between oncogenic signaling and metabolic rewiring. Oncogenic KRAS not only reprograms glucose and glutamine metabolism but also modulates the cellular redox environment, mitochondrial function, and lipid signaling pathways, all converging on GOT2’s multifaceted roles. Understanding these layers of regulation is critical for devising strategies that preemptively address resistance mechanisms that often undermine monotherapies.
The elucidation of GOT2’s role in immune evasion opens additional therapeutic frontiers. By mediating the expression of PTGS2 and other immunosuppressive factors, GOT2 contributes to the shaping of tumor-associated macrophages and the suppression of cytotoxic T lymphocyte infiltration. Therapeutic intervention at this axis could revitalize anti-tumor immunity and overcome immune checkpoint blockade resistance. Thus, GOT2 inhibition might serve as a cornerstone in combinatorial regimens seeking to reinvigorate the immune microenvironment alongside metabolic disruption.
Preclinical evidence supports the notion that targeting GOT2 impairs cancer cell proliferation both by depriving essential metabolic intermediates and by inducing oxidative distress. The consequent DNA damage and cell cycle arrest represent vulnerabilities that can be exploited synergistically with DNA-damaging agents or redox modulators. This multi-dimensional attack on tumor survival pathways renders GOT2 an exceptionally compelling target, deserving of intensified drug discovery efforts.
The microenvironmental dynamics further complicate GOT2 targeting strategies. Crosstalk between pancreatic cancer cells and stromal compartments including CAFs, immune cells, and extracellular matrix components facilitates metabolic symbiosis and resistance. Comprehensive targeting approaches will need to address this ecological network, pairing GOT2 inhibition with therapies that disrupt these cooperative interactions.
In sum, GOT2 embodies a dual threat to pancreatic cancer cell survival: it orchestrates a unique metabolic program vital for bioenergetic and anabolic needs while simultaneously undermining host anti-tumor immunity through transcriptional reprogramming of the tumor microenvironment. This dual functionality heralds a paradigm shift in cancer therapy, whereby metabolic enzymes can no longer be viewed merely as biochemical catalysts but as integral regulators of tumor-host interplay.
As research intensifies, the future of pancreatic cancer treatment may pivot on the successful translation of GOT2 inhibition from bench to bedside. Addressing the challenges of metabolic adaptation and immune suppression concurrently holds promise for breakthroughs in a malignancy long considered intractable. The convergence of structural biology, metabolic biochemistry, and immunology encapsulated by GOT2 research exemplifies the integrated approach necessary for next-generation cancer therapeutics.
Subject of Research: GOT2 enzyme functions and therapeutic targeting in pancreatic cancer
Article Title: GOT2: New therapeutic target in pancreatic cancer
News Publication Date: Not specified
Web References: http://dx.doi.org/10.1016/j.gendis.2024.101370
References: Bu, Jiarui; Miao, Zeyu; Yang, Qing. GOT2: New therapeutic target in pancreatic cancer, Genes & Diseases, Volume 12, Issue 4, 2025, 101370
Image Credits: Genes & Diseases
Keywords: GOT2, pancreatic cancer, glutamine metabolism, malate-aspartate shuttle, mitochondrial enzyme, redox balance, reactive oxygen species, PPARδ, immunosuppression, metabolic reprogramming, oncogenic KRAS, tumor microenvironment
Tags: bioenergetics in cancer proliferationglutamic-oxaloacetic transaminase 2 researchGOT2 as a therapeutic targetKRAS mutations and cancer metabolismmalate-aspartate shuttle in tumorsmetabolic reprogramming in cancermitochondrial enzymes and cancerNAD+/NADH redox balance in cancer cellsnucleotide biosynthesis in pancreatic tumorspancreatic cancer treatment innovationstumor viability and immune evasionunconventional glutamine metabolism in pancreatic cancer