Human Tumor Organoids: Revolutionizing Cancer Modeling and Precision Therapy
Cancer research has experienced a paradigm shift with the advent of human tumor organoids—three-dimensional cultures derived directly from patient tumors that faithfully recapitulate the diverse cellular and molecular characteristics of the original malignancies. Unlike traditional cell lines, tumor organoids preserve patient-specific heterogeneity, making them invaluable tools for investigating complex cancer biology and assessing therapeutic responses with unprecedented fidelity. These dynamic living biosensors provide researchers and clinicians with a powerful platform that bridges mechanistic insights and precision medicine, though challenges remain in translating their full potential into routine clinical use.
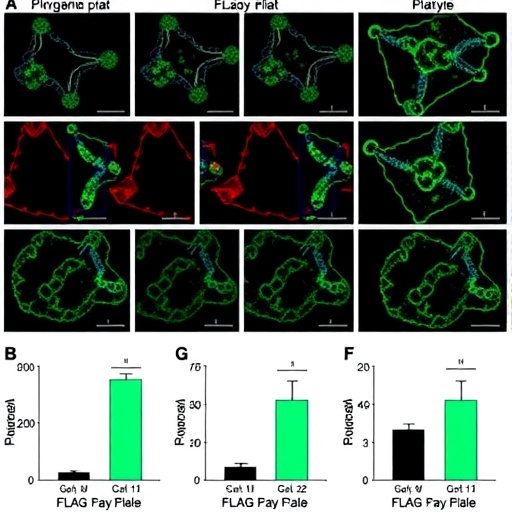
At the core of organoid technology is its ability to maintain the multifaceted tumor microenvironment, including diverse cellular populations and extracellular matrix components that are often lost in simpler in vitro models. Recent advances in culture engineering and biomaterials have been instrumental in stabilizing tumor phenotypes and enhancing the interpretability of drug screening data. Synthetic matrices, decellularized extracellular scaffolds, and scaffold-free culture systems, combined with dynamic perfusion bioreactors, are increasingly deployed to mimic in vivo conditions, ensuring the preservation of critical biophysical and biochemical cues that govern tumor behavior and drug sensitivities.
The tumor microenvironment itself is a complex ecosystem, comprising cancer-associated fibroblasts, immune effectors, vascular networks, and extracellular matrix remodeling enzymes—all of which deeply influence tumor progression and therapeutic resistance. To faithfully reconstruct these intricate interactions, researchers have developed sophisticated co-culture strategies that integrate stromal and immune cells alongside tumor organoids. This holistic approach enables robust modeling of tumor–host interactions and provides a more physiologically relevant context for functional phenotyping, essential for translational applications.
Translation of tumor organoids from bench to bedside is further driven by cutting-edge technologies that enhance scalability and standardization. Organoids-on-chip platforms facilitate precise microenvironmental control and real-time monitoring, while three-dimensional bioprinting enables reproducible generation of complex tissue architectures. High-throughput miniaturized screening combined with multi-omics data integration and machine learning analytics empowers rigorous functional drug-response profiling, accelerating the identification of personalized therapeutic regimens with clinical relevance.
Despite these remarkable advances, the field grapples with critical issues of reproducibility and translatability. Variability in culture protocols and biomaterials can cause divergent tumor states and drug responses, underscoring the necessity for integrated quality control and cross-laboratory standardization. Developing universal benchmarks for organoid phenotypic stability and assay validation remains a paramount goal, as uniformity is indispensable in converting these models into reliable decision-making tools.
Equally pressing are ethical considerations surrounding the sourcing and use of patient-derived tissues. Proper governance frameworks must encompass consent processes, data privacy, and equitable access to emerging therapies informed by organoid platforms. As organoids become increasingly embedded in clinical pipelines, fostering transparent ethical standards will underpin responsible deployment and public trust in this transformative technology.
The living-biosensor framework posited in recent research encapsulates the multifaceted potential of tumor organoids, unifying mechanistic experimentation, microenvironmental recapitulation, and functional drug response into an integrative platform for precision oncology. By situating organoid technology as both a discovery engine and a clinical decision aide, this paradigm offers a practical roadmap from model establishment to therapeutic translation, embodying a new frontier in cancer medicine.
Looking forward, the convergence of biomaterials science, tissue engineering, and computational analytics holds promise to refine organoid systems further, enhancing their physiological relevance and scalability. Multi-disciplinary collaboration will be key to overcoming remaining technical and biological hurdles. Prospective studies harmonizing organoid-derived biomarker discovery with patient outcomes will validate their prognostic and predictive value, ultimately informing tailored treatment regimens that improve survival and quality of life.
In conclusion, tumor organoids represent a transformative leap in cancer research, providing a living, patient-specific platform that recapitulates tumor complexity and enables functional drug testing with clinical fidelity. Through advances in biomaterials, microenvironment reconstruction, and integrative high-throughput technologies, organoids are poised to revolutionize precision therapy. However, realizing their full translational potential demands concerted efforts in standardization, ethical governance, and cross-disciplinary innovation. The future of cancer modeling and individualized treatment is bright, with tumor organoids at its core.
Subject of Research:
Not applicable
Article Title:
Harnessing human tumor organoids for cancer modeling and precision therapy
News Publication Date:
16-Feb-2026
Image Credits:
HIGHER EDUCATION PRESS
Tags: 3D tumor culturesbiomaterials for tumor organoidscancer heterogeneity modelingcancer modeling with organoidsdynamic perfusion bioreactorshuman tumor organoidsorganoid drug screeningpatient-derived cancer modelsprecision cancer therapysynthetic extracellular matricestranslational cancer researchtumor microenvironment in organoids