Advanced microscopy technique opens up inner working of cells
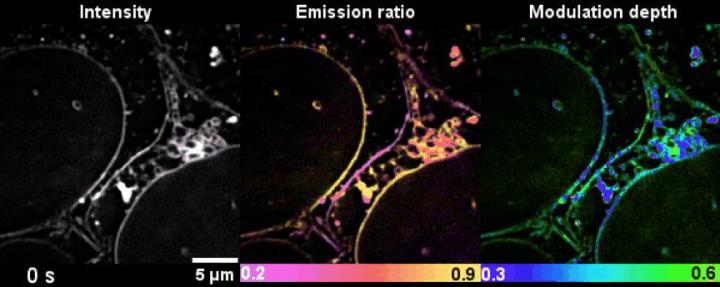
Credit: From Nature Communications Karl Zhanghao et al 2020 (doi.org/10.1038/s41467-020-19747-0)
The ability to “see” the inner workings of structures (organelles) within cells, in real time, offers the promise of advancements in disease diagnosis and treatment. Organelle dynamics drive the self-efficient micro-world of cells, but current super-resolution microscopy techniques used to track these interactions have limitations.
Now an advanced technique called SPOT (Spectrum and Polarization Optical Tomography) is giving researchers a “street view” of the vital lipid membranes surrounding organelles and by so doing opens up the opporutnies to study the sophisticated world of lipid dynamics.
The researchers say it’s a significant development, building on earlier work on super-resolution polarization microscopy.
The research, published in Nature Communications was developed through a collaboration between University of Technology Sydney-Southern University of Science and Technology (UTS-SUStech) Joint Research Centre for Biomedical Materials & Devices, and Peking University.
Lead author Dr Karl Zhangao from UTS-SUSTech Joint Research Centre said that lipid membranes surround most organelles and play a significant role.
“Their shape, composition and phase synergistically regulate biophysical membrane properties, membrane protein function and lipid-protein interactions.”
“However it is challenging to observe such a level of complexity due to their similar chemical composition,” he said.
Simply using one dye that universally stains the lipid membranes, SPOT can simultaneously reveal lipid membrane morphology, polarity, and phase from measuring the intensity, spectrum, and polarization, respectively. Combined with lipophilic probes, the team successfully revealed more than ten types of organelles simultaneously, and their sophisticated lipid dynamics.
Using the new imaging platform established at SUStech, researchers observed the multi-organelle interactive activities of cell division, lipid dynamics during plasma membrane separation, tunneling nanotubules formation, and mitochondrial cristae dissociation.
“This is the first time researchers have been able to quantitatively study the lipid heterogeneity inside subcellular organelles,” senior author Professor Dayong Jin says. Professor Jin is Director of UTS-SUStech Joint Research Centre and Director of UTS Institute for Biomedical Materials and Devices.
“This is a very powerful tool for super-resolution imaging the inner working of each single cells, that will advance our knowledge in understanding how cells function, diagnose when a “factory” or a transportation doesn’t work properly within the cell, and monitor the progression of disease,” Professor Jin said
“With such information it isn’t too big a leap to identify pathways for potential drug treatments, as well as examine their efficacy right on the SPOT” he said.
###
Wenhui Liu, a doctoral student in Tsinghua University, and Meiqi Li, a doctoral student in Peking University are also co-first authors of this work
Media Contact
Marea Martlew
[email protected]
Related Journal Article
http://dx.




