Benzodiazepines, widely prescribed for anxiety, insomnia, and seizure disorders, have long been a staple in clinical medicine. Drugs such as Valium and Xanax are effective in the short term, yet concerns about their long-term impact persist. Increasing evidence suggests that extended use of benzodiazepines may influence inflammatory processes in the body, potentially exacerbating conditions like lung inflammation and inflammatory bowel disease. However, the biochemical and molecular underpinnings behind these effects have remained elusive, challenging researchers to delve deeper into the drug’s multifaceted interactions within human physiology.
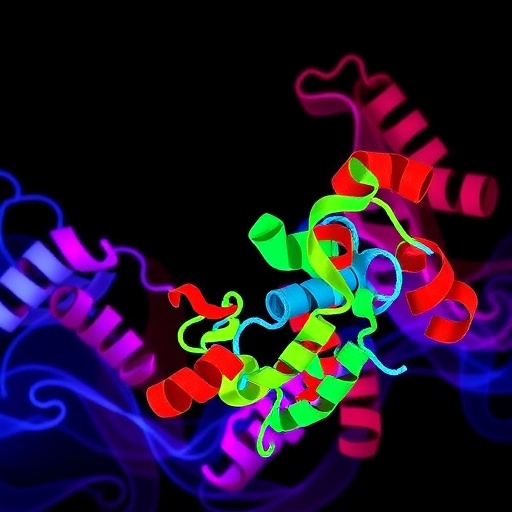
A groundbreaking study spearheaded by Virginia Commonwealth University (VCU) and Columbia University has illuminated the role of a mitochondrial protein, human TSPO1 (HsTSPO1), in benzodiazepine-related inflammation. This membrane-attached protein, found on the outer mitochondrial membrane, has challenged researchers for decades due to its ambiguous structure and unclear function. The results of this research, published recently in the prestigious Proceedings of the National Academy of Sciences, unveil a cholesterol-dependent enzymatic activity of HsTSPO1 that could revolutionize our understanding of benzodiazepine side effects and broader inflammatory disease mechanisms.
At the core of benzodiazepine pharmacology is their modulation of the GABAA receptors in the central nervous system, which underlies their anxiolytic and sedative properties. Yet, benzodiazepines also demonstrate significant binding affinity to HsTSPO1, a protein whose exact biological roles have been debated. Earlier hypotheses proposed that HsTSPO1 primarily facilitates cholesterol transport across mitochondrial membranes, impacting steroid hormone biosynthesis. Nonetheless, researchers Youzhong Guo, Ph.D., and Wayne Hendrickson, Ph.D., posit an alternative enzymatic function, inspired by evolutionary biology insights that suggest a conserved enzymatic role across species from bacteria to humans.
Membrane proteins like HsTSPO1 are notoriously difficult to study due to their complex embedding within lipid bilayers. Traditional approaches using detergents to isolate these proteins often disrupt native lipid interactions critical for stability and function. Overcoming this hurdle, Guo and colleagues engineered a detergent-free system, termed native cell membrane nanoparticles, which preserves the native lipid environment of membrane proteins during analysis. This innovative methodology allowed the team to probe HsTSPO1 with unprecedented precision, revealing its true structural and functional characteristics that had been inaccessible through conventional techniques.
Their investigations show that HsTSPO1 exhibits enzymatic activity by breaking down protoporphyrin IX, a porphyrin crucial to red blood cells involved in oxygen transport. Remarkably, this reaction generates a previously undescribed molecule dubbed bilindigin. Bilindigin appears to regulate reactive oxygen species (ROS) levels, byproducts of cellular metabolism notorious for inducing oxidative stress and inflammatory damage when unchecked. This enzymatic role situates HsTSPO1 as a critical regulator of cellular oxidative balance, implicating it in inflammatory pathways central to benzodiazepine side effects.
This newfound enzymatic function offers a compelling molecular explanation for the inflammatory side effects observed during long-term benzodiazepine treatment. By binding to HsTSPO1, drugs like Valium may inhibit its ability to modulate ROS levels effectively, resulting in increased cellular stress and inflammation. These findings not only deepen the understanding of benzodiazepine pharmacodynamics but also suggest that mitigating such interactions could enhance therapeutic safety profiles.
Beyond benzodiazepines, the implications of this research extend to major chronic diseases characterized by dysregulated oxidative stress and inflammation, such as Alzheimer’s disease, multiple sclerosis, arthritis, and various cancers. HsTSPO1’s critical role in ROS management positions it as an attractive target for novel drug development aimed at these conditions. Tailoring drugs to either avoid HsTSPO1 inhibition or modulate its activity selectively could foster therapeutic breakthroughs in managing these complex disorders.
The study’s success is anchored in the interplay between cutting-edge biochemical techniques and evolutionary theory. The researchers’ hypothesis, that the function of TSPO1 as an enzyme is conserved from prokaryotes to humans, was substantiated by their results. This evolutionary perspective underscores the value of studying ancient protein families to uncover mechanisms relevant to human health and disease.
Guo reflected on the challenges faced by scientists who have long been stymied by the instability of membrane proteins outside their native environment. Their detergent-free approach overcame these limitations, providing a structural and functional portrait of HsTSPO1 interacting with cholesterol and substrate molecules in physiologically pertinent conditions. This breakthrough sets a precedent for how membrane proteins involved in disease processes can be studied more effectively.
Hendrickson highlighted the dual aspects of HsTSPO1’s enzymatic activity: it not only facilitates the breakdown of protoporphyrin IX but also contributes to maintaining cellular redox balance by controlling ROS. This dual functionality may explain the enigmatic nature of HsTSPO1 observed across previous studies. Importantly, it points toward new avenues for drug discovery, where enzyme modulation rather than receptor blockade takes precedence.
Pharmaceutical companies stand to benefit substantially from these revelations. With clearer insight into the molecular interplay between benzodiazepines and HsTSPO1, the design of next-generation anxiolytics can aim for efficacy devoid of ROS-mediated inflammatory liabilities. Moreover, therapeutic innovation directed at HsTSPO1 may yield promising interventions for inflammatory and neurodegenerative diseases that remain challenging to treat.
Ultimately, this research highlights the importance of integrating structural biology, biochemistry, and pharmacology to untangle complex drug mechanisms. As Guo emphasizes, understanding proteins like HsTSPO1 in their near-native environment is vital for translating molecular insights into tangible clinical advancements. The research ushers in a new era where safer benzodiazepine therapy and novel inflammation-targeted drugs can be more intelligently designed.
Benzodiazepines maintain a crucial role in medicine, but with evolving insights into their broader biological interactions, the future points toward refined therapeutics with minimized side effects. The elucidation of HsTSPO1’s enzymatic role marks a significant leap forward, not only in addressing benzodiazepine-associated inflammation but also in opening new therapeutic frontiers for conditions rooted in oxidative stress and chronic inflammation.
Subject of Research: Human TSPO1 protein function and its involvement in benzodiazepine-related inflammation
Article Title: Cholesterol-dependent enzyme activity of human TSPO1
News Publication Date: 27-Mar-2025
Web References: https://www.pnas.org/doi/abs/10.1073/pnas.2323045122
References: DOI: 10.1073/pnas.2323045122
Keywords: Drug research, Discovery research, Chemical structure, Protein structure, Side effects, Drug therapy, Hormone therapy, Psychiatric disorders, Drug design
Tags: anxiety and insomnia medicationsbenzodiazepine pharmacology and GABAA receptorsBenzodiazepine side effectsbenzodiazepines and inflammatory bowel diseasebenzodiazepines and lung inflammationbiochemical mechanisms of benzodiazepinesimpact of benzodiazepines on human physiologyinflammatory processes and benzodiazepineslong-term benzodiazepine usemitochondrial protein TSPO1Proceedings of the National Academy of Sciences studyresearch on benzodiazepine interactions