In a pioneering study that challenges established immunological paradigms, researchers at the University of Massachusetts Amherst have identified a previously unrecognized subset of naïve CD8 T cells endowed with intrinsic memory lineage potential. This discovery not only reshapes our understanding of T-cell biology but also holds tremendous promise for the future of cancer immunotherapy and vaccine development.
T cells, fundamental components of the adaptive immune system, are traditionally understood to operate through a binary fate following antigenic encounter: naïve CD8 T cells either activate and differentiate into effector “killer” T cells or transition into long-lived memory cells after the elimination of a pathogen. The memory cells serve as sentinels, retaining a “molecular diary” of prior infections to enable rapid and robust responses upon re-exposure. The classic dogma has held that naïve T cells are functionally undifferentiated and only commit to these paths following antigen recognition.
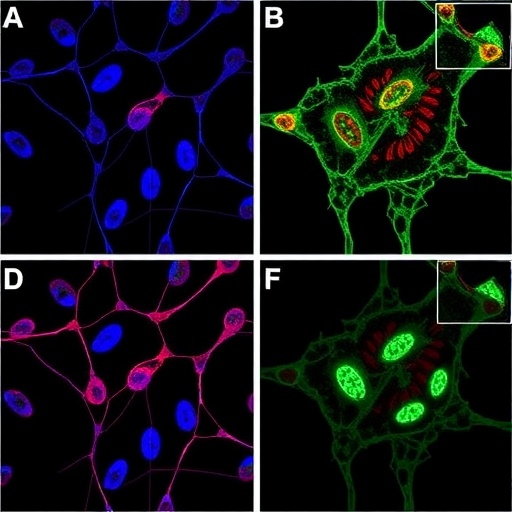
Contrary to this long-standing belief, the UMass Amherst team led by Associate Professor Leonid Pobezinsky uncovered a small population of naïve CD8 T cells that are preordained to differentiate into memory cells. These cells express a unique protein, Dapl1, which serves as a biomarker indicative of their memory-committed fate. Utilizing a sophisticated mouse model engineered to fluoresce upon Dapl1 expression, the researchers meticulously tracked and characterized these elusive cells in vivo.
This subpopulation of Dapl1-positive naïve CD8 T cells exhibits a stem cell-like phenotype, conferring remarkable plasticity and self-renewal capabilities. Such characteristics suggest that these cells can readily give rise to diverse T-cell lineages, adapting dynamically to the immune milieu. The stem-like quality is a critical attribute, as it allows these memory precursors to act as a reservoir, sustaining long-term immunological vigilance.
Microscopic evaluation revealed these antigen-inexperienced T cells residing in a quiescent state yet already expressing molecular signatures indicative of memory lineage commitment. This poised state implies a strategic immune preemption mechanism where certain naïve cells bypass the classical activation pathway, ensuring a rapid and efficient response even without prior exposure.
The implications for cancer immunotherapy are profound. Traditional therapeutic approaches often rely on stimulating the immune system to recognize tumor-specific antigens, a process plagued by issues of variability, inefficiency, and immunosuppressive tumor microenvironments. The discovery of Dapl1+ memory-primed naïve T cells opens up possibilities for designing vaccines and therapies that harness these cells’ intrinsic memory programming to preemptively recognize and combat malignant cells.
Moreover, the research suggests the feasibility of training these pre-wired memory cells in advance, potentially “programming” them against specific oncogenic antigens before tumor emergence. Such a strategy could revolutionize preventive oncology and personalized immunotherapeutics, providing proactive rather than reactive immune defense.
The multidisciplinary approach driving this breakthrough incorporated advanced developmental biology tools pioneered by co-author Professor Dominique Alfandari, who developed antibodies enabling the precise detection and isolation of these memory-committed naïve cells. Parallel efforts by immunologist Wei Cui contributed vital mouse models, reinforcing the functional validity of Dapl1 as a biomarker and establishing a robust platform for future translational research.
While these findings currently derive from murine models, the team is actively investigating whether analogous Dapl1+ naïve memory precursors exist in humans. Confirming their presence in human immune systems could initiate a new era of targeted cancer immunotherapies, using patients’ own immune cells re-engineered ex vivo to heighten anti-tumor efficacy.
This paradigm-shifting work redefines immunological memory formation as a more nuanced and preordained process, integrating novel insights into T-cell ontogeny and functional specialization. It challenges researchers and clinicians alike to re-examine existing frameworks and to envision therapies that exploit this previously unappreciated cellular niche.
As cancer continues to be a formidable global health challenge, innovations that empower the immune system’s natural surveillance machinery are urgently needed. The identification of Dapl1+ memory-lineage predisposed naïve CD8 T cells illuminates a new frontier in immunological research, one where the immune system’s inherent plasticity can be leveraged with unprecedented precision and efficacy.
The research, funded by the National Institutes of Health and the National Research Service Award, underscores the critical importance of interdisciplinary collaboration in driving medical breakthroughs. It serves as a compelling illustration of how converging expertise in immunology, developmental biology, and genetic engineering can yield transformative insights.
In essence, this discovery promises to accelerate the timeline for next-generation vaccines and immunotherapies, offering hope for more effective cancer treatments grounded in the sophisticated biology of T-cell memory. As the field advances, understanding and manipulating these pre-wired memory precursors will become an essential strategy in combating not only cancer but a spectrum of infectious diseases.
Subject of Research: Immunological memory formation in naïve CD8 T cells and implications for cancer immunotherapy
Article Title: A Dapl1+ subpopulation of naïve CD8 T cells is enriched for memory-lineage precursors
Web References:
https://doi.org/10.1126/sciadv.adx5687
Image Credits: UMass Amherst
Keywords: CD8 T cells, immunological memory, naïve T cells, Dapl1 protein, memory-lineage precursors, cancer immunotherapy, stem-cell-like T cells, vaccine development, immunological dogma, T-cell differentiation
Tags: adaptive immune system mechanismscancer immunotherapy advancementsDapl1 protein as a biomarkerimmunological memory in T cellsimmunological paradigms challengeintrinsic memory lineage potential in T cellsmemory cell function in immunitynaïve CD8 T cell differentiationpathogen-fighting cell populationsT cell biology insightsUMass Amherst research breakthroughsvaccine development innovations