In a landmark development that promises to reshape the landscape of targeted cancer therapies, researchers at the University of British Columbia (UBC) Faculty of Medicine have engineered an innovative antibody-drug conjugate (ADC) that exhibits extraordinary precision in identifying and destroying cancer cells, particularly those driven by oncogenic fusions. This groundbreaking therapeutic approach has shown exceptional efficacy in preclinical models, including the formidable Ewing sarcoma, igniting hope for expedited translation into human clinical trials.
The crux of this breakthrough lies in targeting the interleukin-1 receptor accessory protein (IL1RAP), a cell surface antigen selectively overexpressed on malignant cells but strikingly absent from healthy tissues. By conjugating cytotoxic agents to monoclonal antibodies specifically recognizing IL1RAP, the research team has effectively created a molecular delivery system capable of ferrying lethal payloads exclusively to cancerous cells. This strategy sharply contrasts with conventional chemotherapies, which indiscriminately affect normal and malignant cells alike, frequently leading to debilitating side effects.
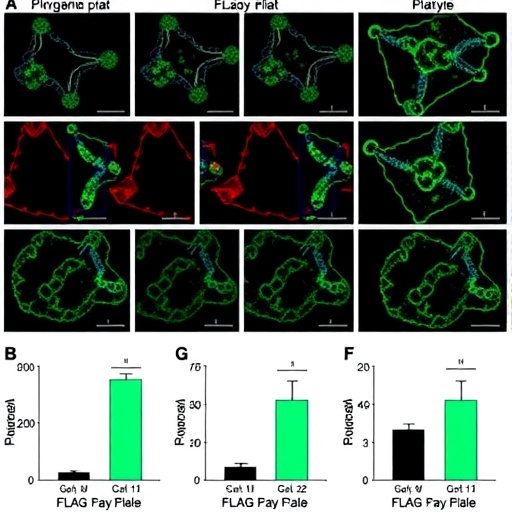
Ewing sarcoma, a rare and aggressively metastatic bone cancer predominantly afflicting children and young adults, has notoriously defied conventional therapies, underscoring an urgent need for innovative treatments. Utilizing sophisticated in vivo and in vitro models, the UBC team demonstrated that their IL1RAP-directed ADC not only eradicated established tumor masses but also significantly mitigated metastatic dissemination. Remarkably, the therapeutic benefits extended beyond Ewing sarcoma, exhibiting potent antitumor activity in lymphoma and other malignancies bearing oncogenic fusions such as NTRK gene rearrangements, underscoring the broad applicability of this approach.
The study, published in the prestigious journal Cancer Discovery, represents an international collaboration spanning multiple continents, blending academic expertise with industrial innovation. Pioneering work first identified IL1RAP as a pivotal facilitator of cancer cell survival in the bloodstream, particularly during the metastatic cascade where tumor cells endure oxidative stress, shear forces, and immune surveillance. This protein acts as a protective shield, enabling malignant cells to colonize distant tissues. By turning this adaptive mechanism into a therapeutic vulnerability, the researchers have ushered in a paradigm shift in cancer treatment.
One of the most compelling features of the IL1RAP ADC is its remarkable safety profile observed in extensive preclinical testing. The selective expression of IL1RAP on cancer cells allows for minimized off-target toxicity, a critical barrier that has historically hampered the clinical success of antibody-based therapeutics. The ADC’s design employs an optimized linker-payload system that ensures the cytotoxic agent remains inactive during systemic circulation, unleashing its full potency only upon internalization into IL1RAP-expressing tumor cells.
This advancement draws on prior foundational studies by Dr. Poul Sorensen and collaborators, including lead author Dr. Haifeng Zhang, who elucidated the role of IL1RAP in facilitating metastasis — the process by which cancer spreads and accounts for the majority of cancer-related mortalities worldwide. The ability to impair metastatic competency by selectively targeting IL1RAP-expressing cells is a testament to the therapeutic’s precision and potential clinical impact.
Clinical translation now appears imminent. With comprehensive toxicology and efficacy data providing robust validation, the investigators are poised to embark on early-phase human trials. Such trials will be vital in confirming the ADC’s safety, optimal dosing, and therapeutic window in patients. If successful, this could herald a new era of precision oncology where genetically defined cancers, especially those driven by oncogenic fusions, can be managed more effectively with targeted interventions minimizing collateral damage to patients.
The molecular engineering underpinning this ADC involves sophisticated bioconjugation techniques to ensure stable yet cleavable linkages between the antibody and drug. This is crucial because premature release of the cytotoxin could lead to systemic toxicity, while insufficient payload release inside the tumor cell could render the therapy ineffective. The ADCs harnessed in this study, including proprietary molecules ADV581-DXd and ADV101, were intricately designed and manufactured through industry collaborations with companies such as Advesya and DualityBio, highlighting the fusion of academia and biotech innovation.
Moreover, the therapeutic potential of IL1RAP targeting transcends cancer type. Given its expression in a spectrum of fusion-positive malignancies, the strategy holds promise not only for pediatric oncology but also for adult cancers characterized by oncogenic drivers that have historically been elusive to targeted therapies. In doing so, it addresses a substantial unmet medical need in the oncology community.
Metastasis remains the principal cause of cancer lethality, largely because disseminated tumor cells adapt unique survival mechanisms that evade conventional treatments and immune detection. By co-opting the IL1RAP axis, this ADC design aims to penetrate the metastatic shield and deliver a cytotoxic strike precisely where it counts, interrupting the lethal march of metastatic progression at its roots.
Overall, this initiative exemplifies the power of translational research—bridging molecular discovery to therapeutic innovation. The selective targeting of IL1RAP not only eradicates primary tumors but also strikes at metastatic disease, potentially revolutionizing outcomes for patients who currently face limited therapeutic options.
In conclusion, the development of IL1RAP antibody-drug conjugates reflects a monumental stride forward for the field of targeted cancer therapy. Integrating molecular biology, antibody engineering, drug conjugation chemistry, and preclinical validation, this precision medicine approach could soon translate into life-saving treatments. With human trials on the horizon, the oncology community eagerly anticipates the outcomes that could redefine cancer care for fusion-driven malignancies across the globe.
Subject of Research: Cells
Article Title: IL1RAP antibody-drug conjugates potently target primary and metastatic disease in multiple oncofusion-driven cancers
News Publication Date: 13-Apr-2026
Web References:
Cancer Discovery Article
DOI Link
Image Credits: Sorensen Lab
Keywords: Cancer treatments, Cancer, Bone cancer, Tumor development, Sarcoma, Metastasis, Oncology, Translational research
Tags: antibody-drug conjugate developmentEwing sarcoma treatment advancesIL1RAP cancer targetinginnovative bone cancer treatmentsmonoclonal antibody drug deliverynovel cancer therapeutic agentsoncogenic fusion-driven cancerspediatric and adult cancer therapiesprecision oncology for sarcomaspreclinical cancer research modelsreducing chemotherapy side effectstargeted cancer therapies