Bioconductive ink uses body’s own electricity to guide nerve cell growth
Credit: RMIT University
Researchers have developed a neuron-growing ink that uses the body’s own electrical signals to precisely guide the growth of nerve cells.
The bioconductive ink can be printed in lines to direct where neurons grow, cracking a major challenge in the emerging field of nerve engineering.
The team of researchers from Australia, India and Bangladesh have tested the ink on a biocompatible scaffold, with their promising lab results published in the journal RSC Advances.
Lead author, RMIT University’s Dr Shadi Houshyar, said concentrating the growth of nerve cells in precisely ordered lines was essential to be able to reconnect nerves and heal traumatic nerve injuries.
“Nerve cells need to be meticulously guided to regrow between the broken ends of a nerve – if they just build up anywhere they will cause more pain or sensory problems,” Houshyar said.
“With our bioconductive ink, we can concentrate the neuron growth where we need it.
“Our research is in early stages but with further development, we hope one day to enable damaged nerves to be fully reconnected, to improve the lives of millions of people worldwide.”
Currently, there are limited options for rebuilding function when an injury results in large peripheral nerve gaps.
Nerve grafts, where surgeons harvest nerves from elsewhere in the body to bridge across a gap, can lead to complications including painful neuromas, misalignment of neural cell growth and injury at the harvest site.
Although emerging alternative techniques such as artificial nerve guides exist, they often fail to achieve full functional or sensory recovery because they don’t properly replicate nerve tissue.
Powering nerve cell regeneration
The new nerve-regenerating ink combines the neurotransmitter dopamine – known to help nerve cell survival – with a conductive carbon nanofibre and polymer.
The nanofibre and polymer enables the controlled release of dopamine from the ink, supporting the survival of developing neurons for longer.
Because it is conductive, the nanofibre can also harness the power of bioelectricity – the electrical signals generated by the nervous system that play a key role in maintaining biological function and can accelerate wound healing.
“Using conductive materials allows free movement of electrons, stimulates cell growth and helps connect injured neural tissue,” Houshyar, a Vice-Chancellor’s Research Fellow in the RMIT School of Engineering, said.
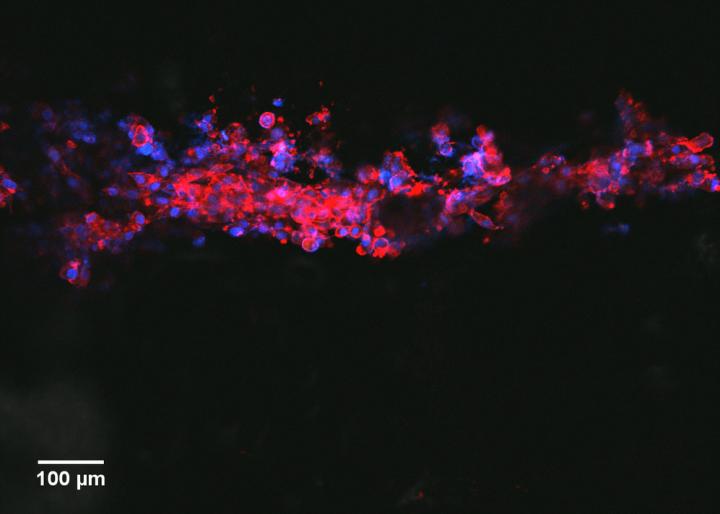
As part of the research, the team also developed a biocompatible scaffold, so the ink could be printed in lines and tested with human cells.
The study found the printed lines supported neural cell attachment and migration – both important for nerve regeneration.
Cell differentiation was also boosted, with the neural cells becoming more specialised as they grew along the lines.
“This supports proper communication with other neurons, which is promising for the establishment of neural circuits for sensory and motor processing – offering hope the technology could lead to a real recovery of nerve function,” Houshyar said.
The next stage for the research is testing the ink and scaffold in pre-clinical animal trials, as well as exploring other applications.
“Our end goal is a nerve engineering solution that can direct the growth of the right nerve cells in the right places,” she said.
“We’re also keen to investigate how we can expand the potential uses of this technology, for speeding up wound healing and improving patient recovery.”
###
The researchers acknowledge the support of the RMIT Microscopy and Microanalysis Facility.
‘Three-Dimensional Directional Nerve Guide Conduits Fabricated with Dopamine Functionalized Conductive Carbon Nanofibre-based Nanocomposite Ink Printing’, with Associate Professor Amitava Bhattacharyya from the PSG Institute of Advanced Studies (India) and collaborators from Jahangirnagar University (Bangladesh), is published in RSC Advances (DOI: 10.1039/d0ra06556k).
Media Contact
Gosia Kaszubska
[email protected]
Original Source
https:/
Related Journal Article
http://dx.




