In a groundbreaking advancement for stem cell biology, researchers have revealed compelling insights into the architectural organization of the genome that governs embryonic stem cell (ESC) pluripotency. The study illuminates the critical role of long-range inter-chromosomal interactions involving the Oct4 distal enhancer loci, a discovery that profoundly expands our understanding of gene regulation in stem cells. This new knowledge not only deepens the molecular narrative of pluripotency maintenance but also opens innovative avenues for regenerative medicine and cellular reprogramming technologies.
Embryonic stem cells are defined by their unique ability to self-renew indefinitely while retaining the capacity to differentiate into any cell type. Central to this remarkable versatility is the delicate orchestration of gene expression networks regulated by key transcription factors, of which Oct4 plays a pivotal role. Oct4 functions as a master regulator of pluripotency; however, the mechanisms by which its activity is spatially and temporally controlled remain partially understood. The current study addresses this critical gap by elucidating the complex genomic interactions at the distal enhancer regions of Oct4.
Utilizing cutting-edge chromatin conformation capture techniques combined with high-resolution imaging, the researchers charted the three-dimensional landscape of the ESC genome. Their findings revealed that the Oct4 distal enhancer loci are not isolated elements but engage in long-range interactions crossing chromosomal barriers. These inter-chromosomal contacts appear to serve as a regulatory hub facilitating coordinated gene expression essential for maintaining pluripotent states.
The novelty of this discovery lies in its challenge to the traditional view that enhancer-promoter communication is predominantly cis-acting, confined within the same chromosome. Instead, the Oct4 enhancers demonstrate a remarkable capacity to physically interact with loci located on different chromosomes, acting in trans to fine-tune gene regulatory networks. This paradigm shift highlights the importance of nuclear architecture and genome organization in modulating ESC identity and functionality.
Moreover, the study details the dynamic nature of these inter-chromosomal interactions, showing them to be highly plastic and responsive to external signaling cues. As ESCs transition towards differentiation, the intensity and frequency of these long-range contacts diminish, signifying their role in sustaining the undifferentiated state. This dynamic regulation underscores an intricate layer of control where spatial genome topology is linked to cellular fate decisions.
Crucially, disturbance or disruption of these long-range enhancer interactions leads to attenuation of pluripotency marker expression and prompts ESC differentiation. This causative evidence points to a direct functional relevance of chromosomal topology in stem cell biology. By manipulating these interactions, it may be possible to modulate stem cell fate, enhancing the precision and efficiency of induced pluripotent stem cell (iPSC) generation and therapeutic cell engineering.
The mechanistic basis underlying these inter-chromosomal communications involves an assembly of architectural proteins and epigenetic modifiers. Proteins such as CTCF and cohesin are implicated in stabilizing these enhancer-promoter loops across chromosomes. Additionally, specific histone modifications enrich the Oct4 enhancer regions, fostering an open chromatin state that facilitates long-distance interactions. Decoding the protein complexes and epigenetic signals involved offers promising targets for pharmacological intervention.
In the broader context, these findings integrate with emerging appreciation for the three-dimensional genome as a fundamental level of genetic regulation. Spatial genome organization influences gene accessibility, transcription factor recruitment, and ultimately cell identity. The ESC genome exemplifies this principle, with its structural organization geared toward maintaining pluripotency through strategic and dynamic chromosomal interactions.
The translational implications of this research are substantial. Understanding enhancer landscapes and their chromosomal crosstalk provides new molecular tools for regenerative medicine. By harnessing or mimicking these genomic configurations, it could become feasible to enhance stem cell culture systems, improve lineage-specific differentiation, and develop novel gene therapies aimed at correcting pluripotency defects.
Furthermore, the study prompts reevaluation of genomic regulatory models in other cell types and developmental stages. The phenomenon of long-range inter-chromosomal enhancer interaction may represent a universal principle in gene regulation, extending beyond ESCs to diverse biological systems and diseases, including cancer. This invites a new frontier of research aimed at mapping chromosomal interactions across the genome and throughout cellular differentiation.
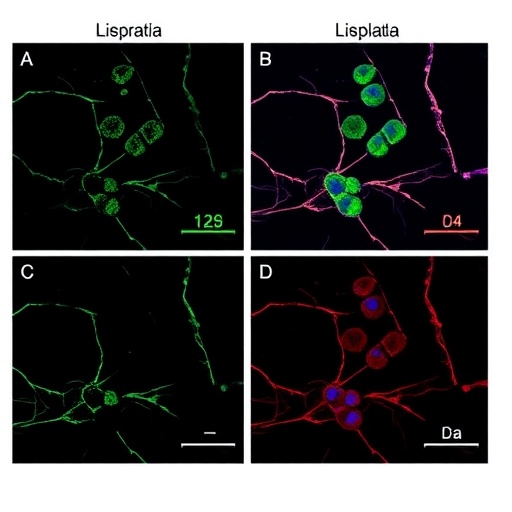
In technical terms, the work employed a refined chromosome conformation capture technique coupled with fluorescence in situ hybridization (FISH), enabling visualization of specific enhancer loci and their interaction partners at unprecedented resolution. The integration of genomic and imaging data provided robust evidence for the spatial proximity of Oct4 enhancers with distinct chromosomal territories, overcoming previous methodological limitations.
Critically, these insights were supported by advanced computational modeling that reconstructed the ESC nuclear topology. This approach elucidated how chromosomal territories are organized and rearranged to facilitate or prevent long-range contacts. Such integrated methodologies underscore the convergent power of multidisciplinary approaches in unraveling complex biological systems.
The revelation of Oct4 distal enhancer inter-chromosomal interaction not only adds depth to stem cell epigenetics but also sets a precedent for investigating other master regulatory genes using similar technological frameworks. This foundational work promises to inspire a cascade of subsequent studies aimed at decoding the genome’s spatial code and its impact on cell fate.
Ultimately, this research enriches our conceptual and practical grasp of cellular identity regulation, paving the way for refined strategies to control stem cell behavior. As the biological sciences continue to embrace the multidimensionality of genomes, discoveries such as these position us closer to mastering cell fate manipulation, with profound implications for health and disease treatment.
Subject of Research:
The study focuses on the role of long-range inter-chromosomal interactions involving the Oct4 distal enhancer loci and their regulatory effects on embryonic stem cells (ESC) pluripotency.
Article Title:
Correction: Long range inter-chromosomal interaction of Oct4 distal enhancer loci regulates ESCs pluripotency.
Article References:
Moon, BS., Huang, D., Gao, F. et al. Correction: Long range inter-chromosomal interaction of Oct4 distal enhancer loci regulates ESCs pluripotency. Cell Death Discov. 12, 183 (2026). https://doi.org/10.1038/s41420-026-02993-4
Image Credits:
AI Generated
Tags: 3D genome architecture in pluripotencycellular reprogramming and enhancer dynamicschromatin conformation capture in stem cellsembryonic stem cell pluripotency regulationenhancer-mediated pluripotency controlESC gene expression networksgenomic organization of ESCsinter-chromosomal enhancer contactsOct4 distal enhancer long-range interactionsregenerative medicine and Oct4 enhancerstem cell self-renewal mechanismstranscriptional regulation by Oct4