Lung cancer remains one of the most formidable adversaries in the realm of oncological diseases, standing as a leading cause of cancer-related mortality worldwide. A significant factor contributing to its lethality is the phenomenon of metastasis, which profoundly affects patient outcomes. Among the myriad of mechanisms that drive cancer progression, metabolic reprogramming has emerged as a critical area of study, particularly the preference of cancer cells for aerobic glycolysis—a metabolic trait known as the Warburg effect. This shift in energy production is not merely a laboratory curiosity; it underpins the aggressive nature of cancer cells and their capacity to survive in hostile environments.
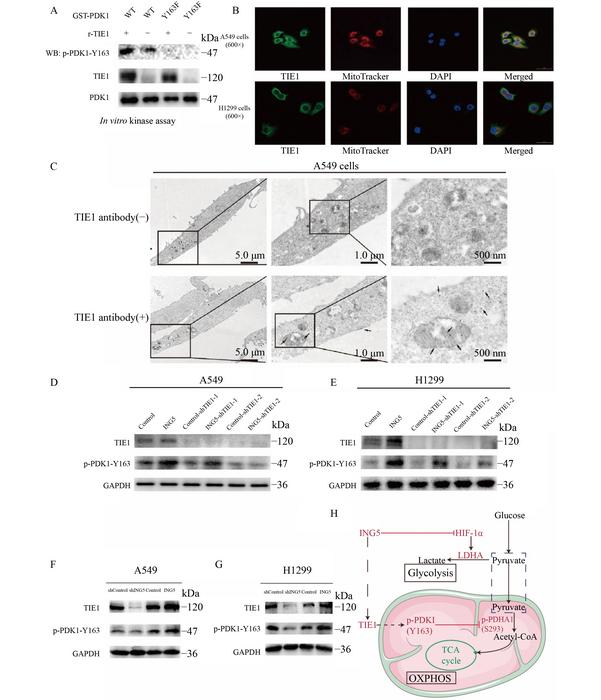
Recent research has illuminated the pivotal role of ING5, a member of the inhibitor of growth (ING) tumor suppressor family. This protein is implicated in the regulation of metabolic pathways in lung cancer cells, providing a promising avenue for therapeutic intervention. The study presents compelling evidence that ING5 acts as a regulator of the Warburg effect, promoting a metabolic environment less favorable for tumor progression. The researchers identified a novel mechanism through which ING5 fosters the phosphorylation of pyruvate dehydrogenase kinase 1 (PDK1) specifically at the tyrosine residue 163 (Y163). This molecular modification plays a crucial role in dampening the Warburg effect, thereby attenuating the malignancy of lung cancer cells.
Delving into the methodologies, the researchers employed a spectrum of experimental approaches, ranging from phospho-proteomics to Western blot analyses and a variety of cellular assays. The combined efforts of these techniques elucidated the relationship between ING5 overexpression and increased PDK1 phosphorylation at Y163. Notably, this phosphorylation event appears to inhibit PDK1’s kinase activity towards its target, the pyruvate dehydrogenase E1 subunit alpha 1 (PDHA1), leading to a cascade of metabolic effects. The culmination of these effects includes a marked decrease in phosphorylation at the S293 site of PDHA1 alongside a promotion of pyruvate dehydrogenase (PDH) enzyme activity. This shift towards enhanced oxidative phosphorylation signifies a profound metabolic transformation favoring survival over aggressiveness.
Moreover, the clinical relevance of PDK1 phosphorylation at the Y163 residue has been further underscored by immunohistochemical analyses of lung tumor tissue microarrays. The data revealed a direct correlation between heightened levels of this specific phosphorylation and improved prognoses in lung cancer patients. Such findings not only reinforce the importance of understanding the intricate metabolic pathways at play within cancer cells but also offer hope for novel prognostic indicators that can aid in patient management.
The implications of PDK1 Y163 phosphorylation stretch beyond mere metabolic reprogramming; they extend into the realms of cellular proliferation and invasion. The research team undertook comprehensive evaluations to grasp the broader impact of this phosphorylation on various malignancy-associated traits. Their findings revealed that the introduction of a phospho-dead mutation (Y163F) drastically negated the beneficial effects induced by ING5 overexpression. This aspect of the research accentuates the essential nature of PDK1 Y163 phosphorylation in maintaining a less aggressive cancer phenotype.
In vivo experiments utilizing mouse xenograft models reinforced the findings established in vitro, showcasing that enhanced expression of ING5 leads to significant reductions in tumor growth and metastasis. The detrimental effects of the Y163F mutation on tumor dynamics further confirm the vital role of this specific phosphorylation event in lung cancer progression. The pathway illuminated by this study presents a breakthrough understanding of the molecular underpinnings that facilitate lung cancer aggressiveness, presenting a potential therapeutic target.
In pursuing the identity of the kinase responsible for mediating PDK1 Y163 phosphorylation, the research brought TIE1 into the spotlight. This receptor tyrosine kinase was observed to be positively regulated by ING5, marking it as a likely player in the metabolic reprogramming of lung cancer cells. Importantly, TIE1 was confirmed to directly phosphorylate PDK1 at Y163, establishing a direct linkage between these molecular entities that could be exploited for therapeutic purposes. Localization studies indicated that TIE1 associates with the mitochondria, a critical site for metabolic processes, further implicating its role in altering cellular energetics.
The implications of targeting the ING5-TIE1-PDK1 signaling axis extend beyond theoretical discussion, inviting consideration of therapeutic strategies that could alter the course of lung cancer treatment. By promoting PDK1 Y163 phosphorylation, researchers may unveil new routes for drug development aimed at reprogramming the metabolism of lung cancer cells, potentially shifting the metabolic landscape towards one less conducive to malignancy. This represents a paradigm shift towards precision medicine that not only targets the cancer itself but also the underlying metabolic imperfections that sustain its growth.
Overall, this groundbreaking research elucidates a multifaceted signaling pathway that underscores the complexity of cancer metabolism. By identifying the critical interactions among ING5, TIE1, and PDK1, this study not only advances our understanding of lung cancer biology but also lays the groundwork for developing novel therapeutic strategies. These findings promise to foster an era of targeted interventions that might improve prognosis and clinical outcomes for lung cancer patients, offering a glimmer of hope in the fight against one of the deadliest cancers known.
As lung cancer research continues to evolve, the exploration of metabolic pathways will undoubtedly remain at the forefront of oncological studies. The insights gleaned from investigating the role of ING5 and its impact on PDK1’s phosphorylation dynamics highlight an urgent need for further research that can translate laboratory findings into tangible clinical applications. Only through a concerted effort among researchers, clinicians, and pharmaceutical developers can we hope to forge a more effective battle plan against lung cancer, a disease that has claimed so many lives.
The intricacies of cancer metabolism are just beginning to be unraveled, but as evidenced by this study, a clearer understanding of these mechanisms is pivotal for developing innovative treatments. By focusing on key molecular players and their interactions, this research not only contributes to our grasp of cancer biology but also inspires the next generation of therapeutic approaches that could save lives.
Subject of Research:
Article Title: ING5 inhibits aerobic glycolysis of lung cancer cells by promoting TIE1-mediated phosphorylation of pyruvate dehydrogenase kinase 1 at Y163
News Publication Date: 14-Sep-2024
Web References:
References:
Image Credits: Credit: Haihua Zhang et al.
Keywords: Lung Cancer, ING5, PDK1, Metabolism, Aerobic Glycolysis, Warburg Effect, TIE1, Pyruvate Dehydrogenase, Oncology, Therapeutics, Clinical Research.