Researchers have developed an innovative laboratory model that promises to revolutionize the study and treatment of myelodysplastic syndrome (MDS), an incurable blood cancer with a high likelihood of progressing into aggressive acute myeloid leukemia (AML). This breakthrough offers a powerful platform that will accelerate the pace of drug discovery and diagnostic development, providing new insights into the molecular mechanisms driving this devastating disease. By utilizing induced pluripotent stem cells (iPSCs) derived directly from patients, scientists have successfully recapitulated the disease process in vitro, allowing them to investigate the genetic mutations responsible for malignant transformation with unprecedented fidelity.
In a landmark study recently published in Nature Communications, a research team led by the University of Birmingham meticulously examined the role of specific genetic alterations in the progression of MDS to AML. Central to their investigation was the gene CEBPA, a transcription factor critical for hematopoietic differentiation. They revealed that mutations disrupting the bZIP domain of CEBPA, especially in the context of coexisting mutations in RUNX1 and SRSF2, act as pivotal drivers of disease progression, reshaping the epigenetic landscape and cellular behavior. This finding underscores CEBPA’s significant oncogenic potential when combined with other mutations within the complex mutational background commonly observed in blood cancers.
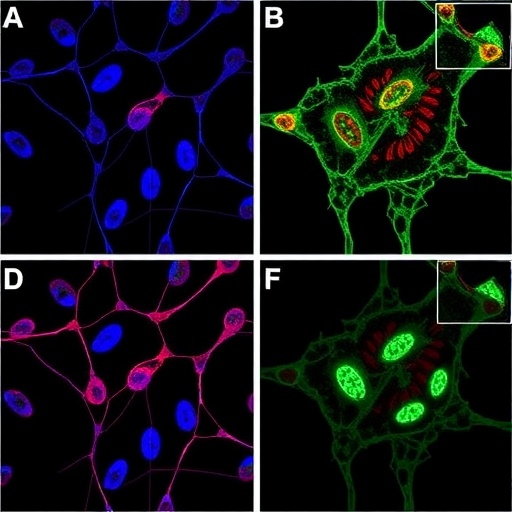
The researchers obtained blood samples from a patient diagnosed with MDS and successfully reprogrammed the patient’s somatic cells into iPSCs through sophisticated genetic reprogramming techniques. These pluripotent cells possess the remarkable ability to differentiate into any cell lineage, providing a versatile tool to model various aspects of hematopoiesis under controlled laboratory conditions. By coaxing these iPSCs to differentiate into both erythroid and myeloid lineages, the team created a robust in vitro system that faithfully mirrored the patient’s original disease state in terms of cellular phenotype and function.
.adsslot_2rl5IeUwXV{width:728px !important;height:90px !important;}
@media(max-width:1199px){ .adsslot_2rl5IeUwXV{width:468px !important;height:60px !important;}
}
@media(max-width:767px){ .adsslot_2rl5IeUwXV{width:320px !important;height:50px !important;}
}
ADVERTISEMENT
Crucially, this approach allowed the researchers to introduce the precise CEBPA mutation identified in the patient two years post-MDS diagnosis, simulating the genetic events underlying disease evolution. This engineered mutation caused a profound shift in cellular dynamics: the production of healthy blood cells was markedly diminished, maturation of white blood cells was blocked, and aberrant cells exhibiting unchecked proliferation emerged. Notably, these malignant cells demonstrated resistance to chemotherapy agents, recapitulating the clinical decline experienced by the patient, thereby validating the physiological relevance of the model.
Analyses of the genetic and epigenetic changes induced by the CEBPA mutation revealed significant alterations in chromatin architecture and gene expression patterns. These changes drove the malignant transformation by reprogramming hematopoietic gene networks and disrupting normal differentiation cues, effectively pushing hematopoietic progenitors onto a leukemogenic trajectory. This highlights the intricate interplay between genetic mutations and epigenetic regulation in the pathogenesis of MDS and AML, demonstrating how a single mutation can have cascading effects on the cellular transcriptional landscape.
The significance of these findings extends beyond elucidating disease biology; they establish iPSC-based models as transformative tools in cancer research. Unlike traditional cell lines or animal models, patient-derived iPSCs provide a human-specific, genetically authentic platform to dissect complex mutational interactions and their impact on disease progression. This can dramatically improve the predictive power of preclinical drug screening assays, enabling more precise identification of therapeutic vulnerabilities inherent in the patient’s genetic context.
Furthermore, the ability to recreate the mutational timeline of MDS progression in vitro offers unparalleled opportunities to develop personalized medicine strategies. By modeling each stage of disease evolution, researchers can evaluate how specific genetic events influence drug responses and identify molecular targets tailored to different phases of malignancy. This could eventually inform clinical decision-making, optimizing treatment protocols to intervene before full-blown leukemia develops.
The study’s senior author, Professor Constanze Bonifer, emphasized the value of integrated genomic and epigenomic profiling in illuminating the ramifications of CEBPA mutation within complex mutational backgrounds. Their comprehensive approach combining cellular phenotyping with high-resolution sequencing technologies provides a blueprint for future research aiming to unravel the multifactorial nature of cancer progression and resistance mechanisms.
Lead author Dr. Paloma Garcia reflected on the promise of the iPSC model system as a new frontier in hematological cancer research. She noted that this methodology not only facilitates the generation of disease-relevant cells but also unlocks the potential for systematic drug screening and functional genomics studies. Such innovative approaches are critical to overcome the historic challenges faced in treating blood cancers that exhibit heterogeneous genetic landscapes and adaptive resistance.
The research team’s openness to collaboration and partnerships underscores the translational potential of their findings. By making this cellular platform accessible to the broader scientific and pharmaceutical communities, they aim to spur the development of next-generation therapeutics that halt disease progression and improve patient outcomes. This collaborative ethos is vital for addressing the urgent unmet medical need posed by aggressive blood malignancies like MDS and AML.
This study represents a pivotal step in integrating cutting-edge stem cell technologies with precision oncology. The insights gained from manipulating patient-specific genetic alterations within iPSC-derived hematopoietic cells pave the way for developing targeted interventions that disrupt malignant transformation pathways. Ultimately, this work advances the quest to transform incurable blood cancers into manageable conditions through tailored, mechanism-based treatments.
By harnessing the power of patient-derived iPSC models, researchers are finally beginning to bridge the gap between clinical observations and molecular underpinnings of blood cancer progression. Their discoveries regarding CEBPA mutation’s definitive role offer a compelling example of how genetic insights can reshape our approach to cancer diagnostics and therapeutics. As this technology matures, it holds the promise to significantly alter the landscape of hematology research and patient care.
Subject of Research: Myelodysplastic syndrome (MDS) progression to acute myeloid leukemia (AML) and the role of CEBPA mutation
Article Title: A heterozygous CEBPA mutation disrupting the bZIP domain in a RUNX1 and SRSF2 mutational background causes MDS disease progression
News Publication Date: 1-Jul-2025
Web References: https://doi.org/10.1038/s41467-025-60192-8
Keywords: Blood cancer, Leukemia, Myeloid leukemia, Animal research
Tags: acute myeloid leukemia progressionCEBPA gene and leukemiadrug discovery platform for cancerepigenetic landscape in blood cancergenetic mutations in MDShematopoietic differentiation and cancerinduced pluripotent stem cells in oncologyinnovative laboratory model for blood cancermolecular mechanisms of myelodysplastic syndromemyelodysplastic syndrome researchNature Communications study on blood cancerRUNX1 and SRSF2 mutations