A new iron catalyst helps preferentially reduce nitric oxide to hydroxylamine, opening doors to pollution control and clean energy.
Credit: Gwangju Institute of Science and Technology (GIST)
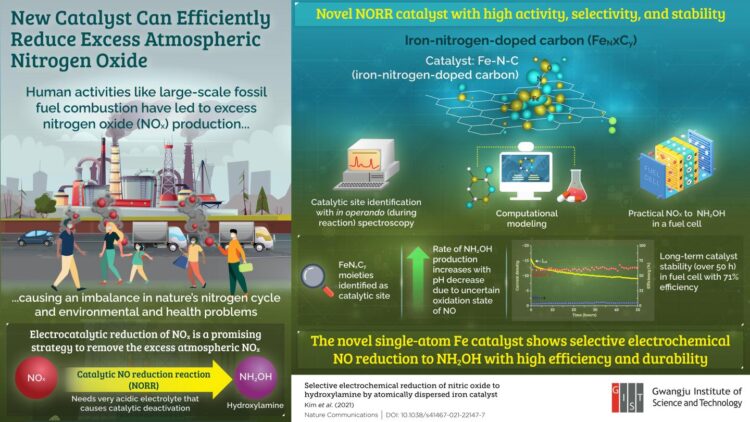
Our reliance on fossil fuels as a primary energy source has pushed air pollution to an all-time high, resulting in several environmental and health concerns. Among the major pollutants, nitrogen oxide (NOx) accumulation can cause severe respiratory diseases and imbalance in the Earth’s nitrogen cycle. Reducing NOx accumulation is, therefore, an issue of utmost importance.
Recently, the conversion of NOx into harmless or even useful nitrogen products has emerged as a promising strategy. Particularly appealing to scientists is the reduction of NOx to hydroxylamine (NH2OH), which can be utilized as a renewable source of energy.
The “make-or-break” step that determines the formation of hydroxylamine is the catalytic electrochemical reduction of nitric oxide (NO), which can either yield hydroxylamine or nitrous oxide (N2O), depending on the electrolyte pH and electrode potential. Studies show that for hydroxylamine formation to dominate over N2O formation, very acidic electrolytes with a pH less than 0 are required. However, such a harshly acidic environment rapidly degrades the catalyst, limiting the reaction. “The development of a new catalyst with high activity, selectivity, and stability is the next challenge,” says Prof. Chang Hyuck Choi from the Gwangju Institute of Science and Technology (GIST) in Korea where he works on the catalysis of electrochemical reactions.
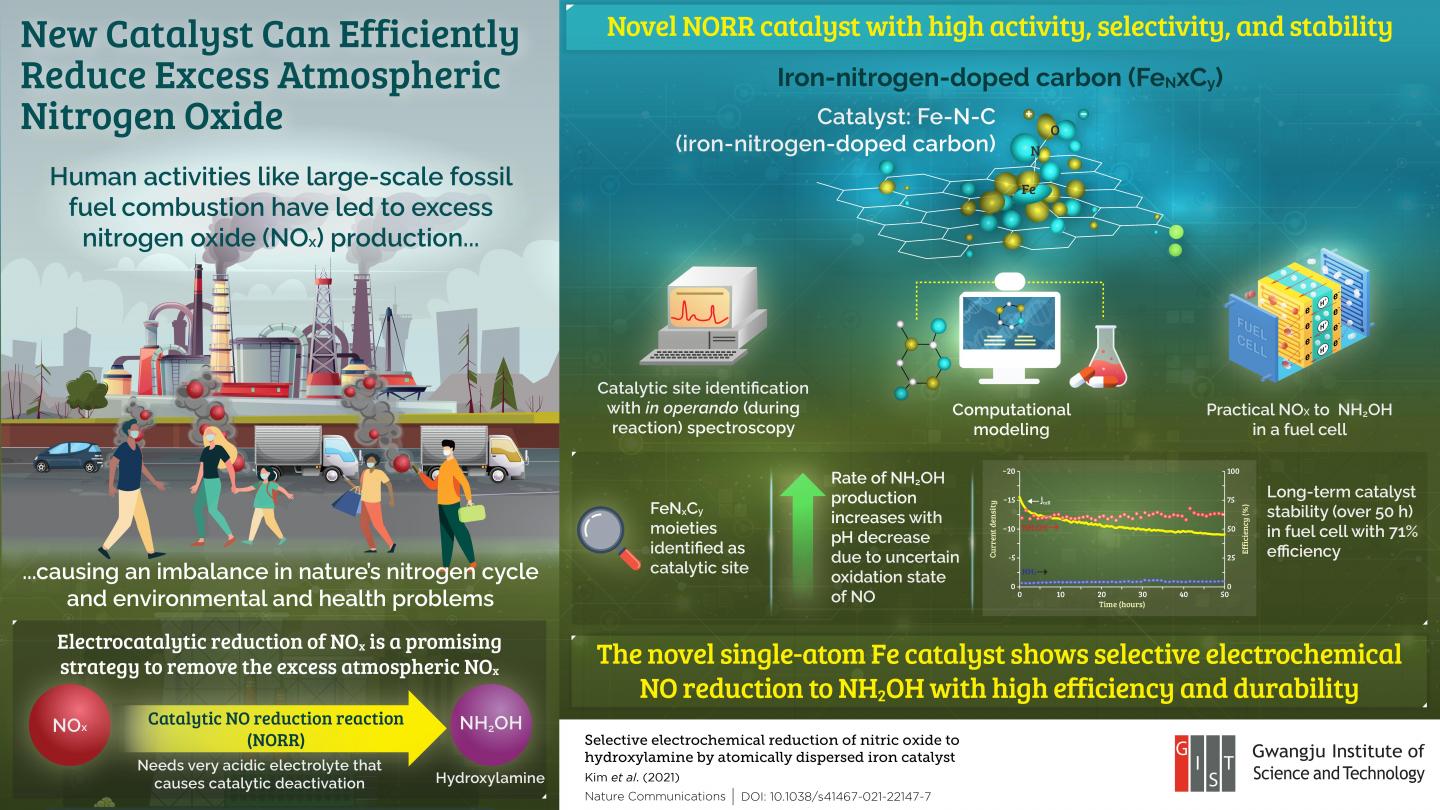
In a recent study published in Nature Communications, Prof. Choi and his colleagues from Korea and France investigated NO reduction in the presence of a new iron-nitrogen-doped carbon (Fe-N-C) catalyst made of isolated FeN
The team performed in operando (i.e., during the reaction) spectroscopy and electrochemical analysis of the catalyst to determine its catalytic site and the pH dependence of NH2OH production.
They identified the active site of the catalyst as the ferrous moieties bonded to the carbon substrate where the rate of NH2OH formation showed a peculiar increase with decreasing pH. The team attributed this peculiarity to an uncertain oxidation state of NO. Finally, they achieved efficient (71%) NH2OH production in a prototypical NO-H2 fuel cell, establishing the catalyst’s practical utility. Moreover, they found that the catalyst exhibited long-term stability, showing no signs of deactivation even after operating for over 50 hours!
The approach not only reduces harmful air pollutants, but also provides a useful byproduct that may find use in ushering in a renewable energy society. “Apart from the applications of hydroxylamine in the nylon industry, it can also be used as an alternative hydrogen carrier. Thus, the new catalyst will not only help reduce the amount of NOx pollutants in our atmosphere but also lead us to a renewable energy future,” Prof. Choi explains.
We can breathe easy knowing that the team’s findings take us a few steps closer to a pollution-free renewable energy society.
###
Reference
Title of original paper: Selective electrochemical reduction of nitric oxide to hydroxylamine by atomically dispersed iron catalyst
Journal: Nature Communications
DOI: https:/
About Gwangju Institute of Science and Technology (GIST)
Gwangju Institute of Science and Technology (GIST) is a research-oriented university situated in Gwangju, South Korea. One of the most prestigious schools in South Korea, it was founded in 1993. The university aims to create a strong research environment to spur advancements in science and technology and to promote collaboration between foreign and domestic research programs. With its motto, “A Proud Creator of Future Science and Technology,” the university has consistently received one of the highest university rankings in Korea.
Website: http://www.
About the author
Prof. Chang Hyuck Choi is an Associate Professor in the School of Materials Science and Engineering at the Gwangju Institute of Science and Technology (GIST) in Korea. He received his B.S. (2007) and Ph.D. (2012) degrees from the Department of Chemical Engineering at the Korea Advanced Institute of Science and Technology (KAIST) in Korea. He was a postdoctoral fellow at KAIST from 2012 to 2014. From 2014 to 2016, he worked as a Humboldt Research Fellow at the Max-Planck-Institut für Eisenforschung (MPIE) in Germany. His research interests mainly focus on the fundamental principles of electrocatalysis for energy conversions.
Media Contact
Nayeong Lee
[email protected]
Original Source
https:/
Related Journal Article
http://dx.