In the ever-evolving landscape of cancer metabolism, new insights often challenge long-standing paradigms about how malignant cells sustain their aggressive proliferation. Emerging research from Van Andel Institute (VAI), recently published in Nature Metabolism, reveals an alternative biochemical pathway cancer cells employ to fuel their growth. This revealing study unpacks the complexity of nutrient utilization in cancer biology, uncovering a non-canonical metabolic route whereby cancer cells convert the ketone body β-hydroxybutyrate (β-OHB) into cytosolic acetyl-CoA — a pivotal molecular precursor integral to lipid synthesis and cell proliferation.
Traditionally, cancer metabolism research has revolved heavily around glucose as the principal substrate fueling tumor growth, framed by the celebrated Warburg effect. Cancer cells preferentially consume glucose and rely predominantly on glycolysis for energy production, even under oxygen-rich conditions. However, this new research complicates this view by demonstrating that cancer cells possess metabolic plasticity that enables them to exploit alternate fuel sources such as ketone bodies, especially β-OHB, which is typically elevated during fasting or carbohydrate-restricted states.
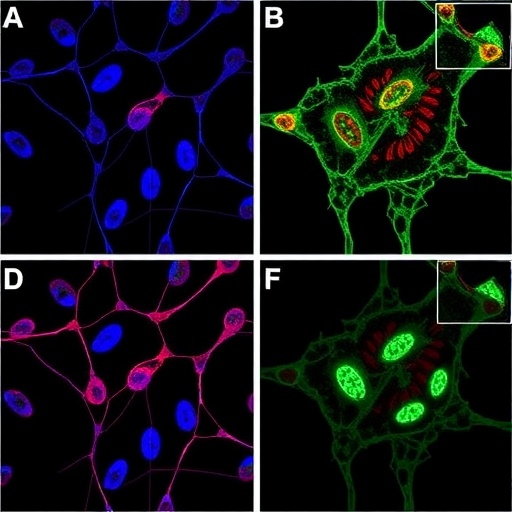
Dr. Evan Lien and his team at VAI explored this metabolic flexibility with meticulous biochemical and molecular investigations. They uncovered that β-OHB does not simply serve as a backup nutrient in situations of glucose scarcity; rather, cancer cells utilize a distinct enzymatic route — diverging from canonical mitochondrial oxidation — to transform β-OHB into acetyl-CoA within the cytosol. This acetyl-CoA pool directly contributes to the synthesis of fatty acids and cholesterol, compounds vital for assembling cellular membranes and supporting rapid proliferation rates characteristic of malignant growth.
The discovery challenges the simplistic notion that glucose is invariably the dominant nutrient in cancer cell metabolism, showing that even in glucose-replete conditions, alternative substrates fuel key biosynthetic pathways. This metabolic versatility allows tumor cells to adapt dynamically to fluctuating systemic nutrient availability, pointing to a multi-layered metabolic resilience that likely contributes to treatment resistance and tumor progression.
Central to this alternative pathway is a specialized enzymatic machinery that departs from the recognized mitochondrial β-oxidation of ketone bodies. Instead, β-OHB enters a cytosolic metabolic circuit involving enzymes that furnish acetyl-CoA without passing through the classical canonical routes. The precise enzymology and regulatory mechanisms orchestrating this non-canonical metabolism have been the focus of the study and offer new biological insights into cancer metabolism, expanding the conceptual framework beyond glucose-centric models.
Interestingly, this study dovetails with recent findings from other VAI laboratories revealing that ketone metabolism is not unique to cancer cells. Immune cells, specifically T lymphocytes, also employ ketones to meet their energetic and biosynthetic demands. Led by Dr. Russell Jones, co-author of the current study, prior research demonstrated T cells’ preference for ketones as an alternative substrate over glucose, endowing them with metabolic backup plans that enhance their anti-cancer functions. The identification of overlapping metabolic strategies between cancer cells and immune effectors suggests a complex metabolic interplay in the tumor microenvironment that may influence therapeutic outcomes.
The broader implications of these findings touch upon the contentious dialogue surrounding ketogenic diets and cancer. While ketogenic regimens have garnered popular attention for their metabolic influence and potential links to cancer management, this research refrains from drawing direct correlations. Dr. Lien emphasized that the study’s scope was confined to elucidating intracellular metabolic pathways rather than dietary interventions, urging caution against simplistic cause-effect conclusions between ketogenic diets and tumor growth.
This expanded understanding of how cancer cells harness multiple nutrients and metabolic routes to sustain acetyl-CoA availability opens fresh avenues for therapeutic exploration. Targeting enzymes exclusive to this alternative β-OHB metabolizing pathway could starve tumors of key biosynthetic precursors without disrupting normal cellular metabolism that depends on canonical pathways. This selectivity could translate into novel treatments with improved efficacy and reduced toxicity profiles.
Moreover, appreciating this multiplicity in fuel utilization underlines the necessity of a nuanced perspective when developing anti-cancer strategies. Combating tumor metabolism may demand combinatorial approaches that simultaneously obstruct several nutrient pathways or modulate systemic metabolic states that influence substrate availability to tumors.
The study adds to a growing consensus recognizing cancer metabolism as a highly adaptable, context-dependent network rather than a fixed program. This metabolic plasticity endows tumors with survival advantages in heterogeneous microenvironments and likely underlies many instances of therapeutic resistance observed clinically. By illuminating previously unappreciated routes of acetyl-CoA synthesis, the research offers critical steps toward decoding this complexity.
Future work will be essential to map the regulatory controls governing this non-canonical ketone utilization and to ascertain its prevalence across cancer types. Investigating how systemic conditions such as fasting, carbohydrate restriction, or metabolic diseases modulate these pathways could further contextualize their role in tumor biology. Additionally, the relationship between tumor ketone metabolism and immune cell function in situ presents an intriguing frontier for exploration, potentially uncovering metabolic cross-talk that shapes tumor immunity and therapy responses.
In summary, the discovery of an alternative β-hydroxybutyrate metabolic pathway supporting cytosolic acetyl-CoA synthesis in cancer cells profoundly expands the biochemical repertoire underpinning tumor metabolism. It compels a re-examination of nutrient utilization paradigms in oncology and highlights the intricate biochemical adaptations that sustain cancer cell survival. As we unravel these metabolic intricacies, new therapeutic vulnerabilities may emerge, paving the way for innovative treatments to disrupt cancer’s metabolic lifelines.
Subject of Research: Cancer cell metabolism; ketone body utilization; β-hydroxybutyrate metabolic pathways.
Article Title: An alternative route for β-hydroxybutyrate metabolism supports cytosolic acetyl-CoA synthesis in cancer cells
News Publication Date: September 8, 2025
Web References:
Van Andel Institute: http://www.vai.org/
Nature Metabolism article DOI: http://dx.doi.org/10.1038/s42255-025-01366-y
References:
Faith C. Kaluba, Thomas J. Rogers, Yu-Jin Jeong, Rachel J. House, Althea Waldhart, Kelly H. Sokol, Samuel R. Daniels, Cameron J. Lee, Joseph Longo, Amy Johnson, Vincent J. Sartori, Ryan D. Sheldon, Evan Lien, Russell Jones et al. “An alternative route for β-hydroxybutyrate metabolism supports cytosolic acetyl-CoA synthesis in cancer cells.” Nature Metabolism, 2025.
Image Credits: Courtesy of Van Andel Institute
Keywords: Cancer research, Metabolites, Metabolism, Metabolomics
Tags: alternative metabolic pathways in cancerbiochemical investigations in oncologycancer metabolism researchglucose and glycolysis in cancerinsights into malignant cell growthketone bodies as fuel sourceslipid synthesis in cancer cellsmetabolic plasticity of tumorsnon-canonical metabolic routestumor proliferation mechanismsVan Andel Institute cancer studyβ-hydroxybutyrate in cancer growth