Expanded use and potential misuse of botulinum neurotoxins increases health risks
Credit: UCI School of Medicine
Irvine, Calif. – February 27, 2020 – New study reveals potential for developing novel antibody-based antitoxins against botulinum neurotoxins (BoNTs), including the most commonly used, yet most toxic one, Botox.
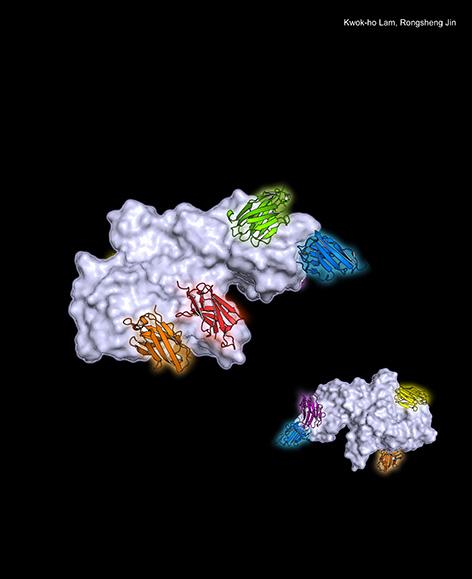
Published in Cell Reports, the paper is titled, “Structural insights into rational design of single-domain antibody-based antitoxins against botulinum neurotoxins.” Led by Rongsheng Jin, PhD, a professor in the Department of Physiology & Biophysics at the University of California, Irvine, School of Medicine, this paper describes how the team first identified the neutralizing epitopes of six anti-BoNT nanobodies (VHHs) based on their crystal structures, then harnessed the structural findings to rationally design bifunctional nanobodies. Different than ordinary nanobodies, bifunctional nanobodies are composed of two nanobodies that bind simultaneously to the toxins.
Based on a mouse model, their findings revealed the bifunctional nanobodies protected mice with much greater potency than the simple combination of two nanobodies.
“In a nutshell, we establish a platform for structure-based rational design of bifunctional antitoxins against BoNTs,” said Kwok-ho Lam, the first author and a project scientist in the Jin lab. “BoNTs can be misused as a bioweapon and thus have been classified as Tier 1 select agents by the Centers for Disease Control and Prevention, which is why there is urgent need for antitoxins.”
Ironically, Botox is a type A botulinum neurotoxin (BoNT/A), and just one of the many different types of botulinum neurotoxins. BoNT/B is another botulinum neurotoxin approved for therapeutic uses, and yet another type, BoNT/E, is in clinical trials.
“Currently, the only available antitoxin remedies are polyclonal antibodies from horse or human serum, which have known health risks and are in limited supply. Monoclonal antibodies are still under development,” said Jin. “And, while it isn’t necessarily a cause for worry, the increasingly popular therapeutic uses of BoNT products also create risks of possible botulism resulting from the medical treatments where they are used.”
###
This study was funded in part by the National Institute of Allergy and Infectious Diseases, the Defense Threat Reduction Agency–Chemical Biological Defense Therapeutics, and the National Institute of General Medical Sciences.
About the UCI School of Medicine
Each year, the UCI School of Medicine educates more than 400 medical students, and nearly 150 doctoral and master’s students. More than 700 residents and fellows are trained at UCI Medical Center and affiliated institutions. The School of Medicine offers an MD; a dual MD/PhD medical scientist training program; and PhDs and master’s degrees in anatomy and neurobiology, biomedical sciences, genetic counseling, epidemiology, environmental health sciences, pathology, pharmacology, physiology and biophysics, and translational sciences. Medical students also may pursue an MD/MBA, an MD/master’s in public health, or an MD/master’s degree through one of three mission-based programs: the Health Education to Advance Leaders in Integrative Medicine (HEAL-IM), the Leadership Education to Advance Diversity-African, Black and Caribbean (LEAD-ABC), and the Program in Medical Education for the Latino Community (PRIME-LC). The UCI School of Medicine is accredited by the Liaison Committee on Medical Accreditation and ranks among the top 50 nationwide for research. For more information, visit som.uci.edu.
Media Contact
Anne Warde
[email protected]
949-824-6357
Original Source
http://som.
Related Journal Article
http://dx.