Credit: Kanazawa University
[Background]
Nonalcoholic steatohepatitis (NASH) is associated with liver inflammation in patients with nonalcoholic fatty liver disease (NAFLD), and it can progress to liver fibrosis at an advanced stage, and also to hepatocellular carcinoma (HCC) and portal hypertension. Although liver fibrosis is considered to be accurately diagnosed via biopsy, noninvasive methods are preferable. Aldo-keto reductase family 1 member B10 (AKR1B10) is an enzyme that is highly expressed in various cancers, including HCC, and it has also been found in advanced liver diseases such as NASH. AKR1B10 is secreted into the stroma from liver cells by binding to heat shock protein 90 (Hsp90) and it can be analyzed in the serum as an index of advanced liver disease. Additionally, AKR1B10 is a cancer-related marker that is correlated with retinoic acid metabolism.
[Results]
Eighty-five NASH patients who were diagnosed via liver biopsy at Kanazawa University Hospital from 2006 to 2017 were enrolled into this study. These patients mostly underwent serum analysis within 1 month before or after the histological diagnosis of NASH. The observation period lasted for 8 years after NASH diagnosis or HCC incidence. The HCC staging of 36 patients was as follows: stage I, two patients; stage II, 18 patients; stage III, 12 patients; and stage IV, 4 patients. Participants were classified into two groups based on advanced Brunt stage 4 (cirrhosis) or other stages.
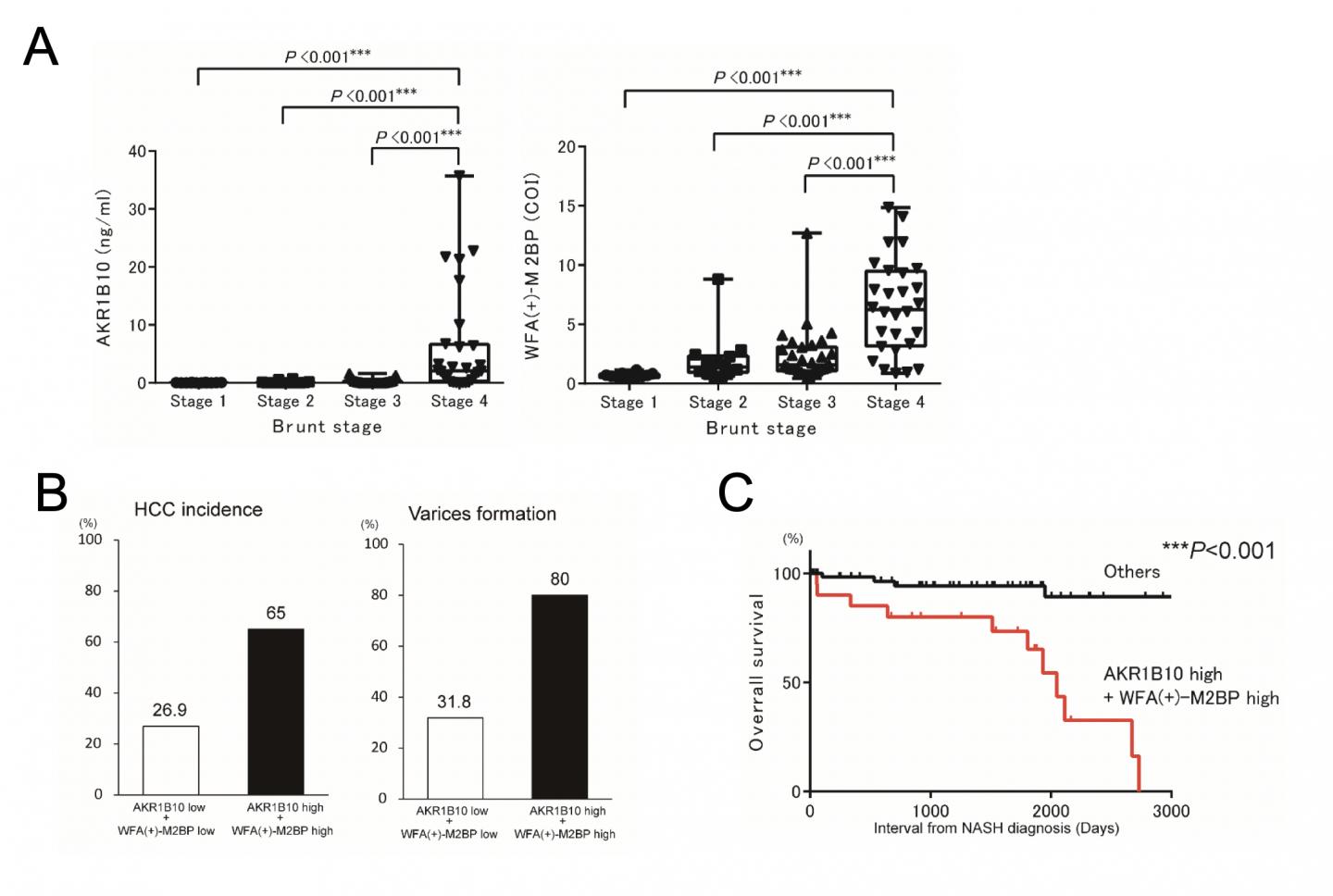
The increase in serum AKR1B10 was significantly associated with an advanced liver fibrosis stage, particularly stage 4 of the Brunt classification (Figure A). The average serum AKR1B10 level was 0.03 ng/mL in stages 1-3 and 2.10 ng/mL in stage 4. At a cutoff of 1.03 ng/mL for stage 4, the sensitivity was 71.4% and specificity was 94.7%. The area under the receiver operating characteristic (AUROC) curve was 0.858. These data indicate that serum AKR1B10 exhibited very high specificity for advanced NASH. Moreover, to confirm that this protein is derived from liver tissues, we performed immunohistochemical staining of the patients’ background liver samples, and we found that patients with higher serum AKR1B10 levels had positive AKR1B10 staining (Figure 1). These results suggested that AKR1B10 protein originated in the liver tissue and was secreted into the blood.
We compared NASH Brunt stage 4 status with other serum liver fibrosis markers and found that serum Wisteria floribunda agglutinin-positive Mac-2 binding protein (WFA(+)-M2BP) (M2BPGi) also showed high sensitivity and specificity although it was also elevated in some stage 2-3 patients, unlike AKR1B10 (Figure A). Thus, we speculated that WFA(+)-M2BP was also a useful marker for predicting advanced NASH fibrosis and that its combination with serum AKR1B10 might provide a powerful diagnostic method.
Using both serum AKR1B10 and WFA(+)-M2BP levels, we analyzed life-threatening events such as HCC incidence and gastroesophageal varix formation in NASH patients. We divided groups based on the AKR1B10 cutoff value (1.03 ng/mL) and the WFA(+)-M2BP value (3.11 cutoff index (C.O.I.)) for Brunt stage 4. The Brunt stage 4 ratio significantly differed between the two groups examined, as follows: high AKR1B10 and WFA(+)-M2BP versus low AKR1B10 and WFA(+)-M2BP. A significant difference was also seen in the HCC incidence ratio and gastroesophageal varix formation ratio (Figure B). Moreover, we found that overall survival after a NASH diagnosis was worse in patients with higher levels of both markers (Figure C). These results indicate that combination analysis using serum AKR1B10 and WFA(+)-M2BP can predict the HCC incidence and portal hypertension associated with a poor prognosis of patients with NASH.
[Significance and future prospects]
There is an inherent advantage in the combination of serum AKR1B10 and WFA(+)-M2BP, which reflects liver fibrosis in NASH, with another factor that can predict an advanced stage or fatal complication of NASH. The utility of these serum markers can facilitate accurate NASH diagnosis and progression.
###
Media Contact
Tomoya Sato
[email protected]
Related Journal Article
http://dx.