The acquisition of totipotency represents one of the earliest and most pivotal events in mammalian development. This remarkable biological transition is defined by zygotic genome activation (ZGA), a process that initiates the embryo’s own transcriptional machinery, and is accompanied by the transient expression of a unique repertoire of 2-cell-stage (2C) specific genes and endogenous retrotransposons such as MERVL. Despite the centrality of this transition, the precise molecular mechanisms by which master transcription regulators orchestrate the sweeping chromatin reconfigurations necessary for totipotency have long eluded clear elucidation.
Emerging from these mysteries, recent groundbreaking research has illuminated a remarkable biophysical process underpinning this fundamental developmental leap: the phase separation behavior of Double Homeobox (DUX) family proteins. These proteins—mouse Dux and human DUX4—have been previously characterized as transcriptional drivers of the totipotent state. However, the mechanistic insights now reveal that beyond simple DNA binding, DUX proteins engage in liquid-liquid phase separation (LLPS) within the nucleus, forming dynamic biomolecular condensates that serve as essential architectural hubs orchestrating genome-wide reprogramming.
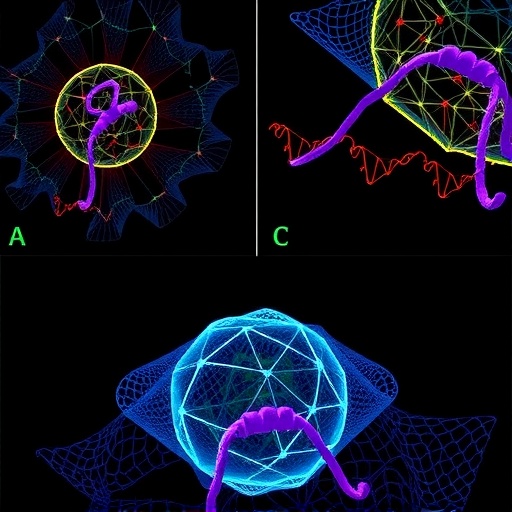
At the core of this phenomenon are intrinsically disordered regions (IDRs) embedded within the highly conserved Homeobox domains of DUX proteins. These disordered motifs, particularly the critical arginine residues (notably R70 and R72 in mouse Dux; R71 and R73 in human DUX4), facilitate multivalent interactions necessary to drive phase separation. This results in the assembly of nuclear condensates—membraneless compartments characterized by liquid-like properties that concentrate transcriptional regulators, cofactors, and chromatin architectural proteins into functionally specialized domains.
These DUX condensates selectively recruit co-activators such as the histone acetyltransferases CBP/p300, as well as architectural proteins like CTCF, known to shape three-dimensional chromatin topology. Intriguingly, phase separation-deficient mutants of DUX, despite retaining normal DNA-binding affinity and continued interaction with co-activators, display a complete loss of function in activating key totipotency markers such as MERVL. This observation underscores the indispensable role of the dynamic, multivalent condensate environment in orchestrating effective transcriptional activation within the reprogramming nucleus.
A pivotal insight from this work is the discovery that DUX phase separation facilitates the de novo assembly of totipotency-specific super-enhancers (SEs). Super-enhancers are extended clusters of enhancers characterized by exceptionally high transcriptional activity that coordinate robust gene expression programs. Utilizing high-resolution Hi-C chromatin conformation capture analyses, the research delineates how DUX condensates actively reshape the 3D genome architecture, inducing pronounced shifts in topologically associating domain (TAD) boundaries and engendering novel enhancer-promoter chromatin loops that were previously absent.
This spatial reorganization transcends traditional cis-regulatory logic by enabling DUX to activate a broad network of 2C-stage genes, including those distal and lacking direct DNA-binding sites for DUX itself. The phase-separated condensates thereby act as nucleation points for complex long-range chromatin interactions, effectively ‘rewiring’ transcriptional networks essential for totipotency programming. This multilayered regulatory paradigm reveals the extent to which biomolecular phase transitions contribute to the fundamental wiring of gene expression during mammalian development.
Functional validation through mutant analyses highlights the biological significance of DUX phase separation. Dux mutants deficient in condensate formation fail to establish the full 2C transcriptome and are severely compromised in their ability to contribute to extraembryonic lineages in chimeric embryo assays, definitively linking phase separation to developmental potency. The failure to induce critical gene networks in these mutants exemplifies how LLPS-driven genome reorganization is not simply a biochemical curiosity but a requisite feature of early embryogenesis.
Extending these findings beyond normal development, the study also explores the pathological implications of DUX4 phase separation in human disease. Facioscapulohumeral muscular dystrophy (FSHD), a debilitating genetic disorder characterized by inappropriate expression of DUX4 in skeletal muscle, is shown to depend on the condensate-forming capacity of DUX4. The phase separation behavior appears to be a key driver of myotoxicity, suggesting that dysregulated LLPS mechanisms underlie the disease’s molecular pathology. This novel insight opens promising therapeutic avenues aimed at modulating phase separation processes to ameliorate or prevent disease phenotypes.
The delineation of DUX family protein phase separation as a driver of 3D genome reprogramming marks a paradigm shift in our understanding of developmental biology. It situates LLPS as a central organizing principle that integrates transcriptional activation with spatial chromatin architecture, enabling totipotent-like states to arise from the zygotic genome. By bridging structural biophysics with gene regulation, this research uncovers a dynamic and adaptable framework through which developmental potency is established and maintained.
Importantly, the molecular features governing DUX phase separation—such as the critical arginine-rich IDRs—offer tangible molecular targets for pharmacological intervention. In the context of developmental biology, synthetic modulation of these phase separation dynamics could allow controlled manipulation of cell potency states, with broad implications for regenerative medicine and cellular therapies. Concurrently, the pathological insights into FSHD underscore LLPS as a potential molecular target in disease contexts, where aberrant condensate formation disrupts cellular homeostasis.
Technological advancements were pivotal in this discovery, including sophisticated live-cell imaging techniques to visualize condensate dynamics, proteomic approaches to identify interacting partners recruited to DUX condensates, and integrative epigenomic mapping to define SE landscapes. The integration of Hi-C genome architecture analyses was particularly transformative, revealing how LLPS-driven molecular assemblies translate into macroscopic changes in nuclear organization that underpin comprehensive transcriptional rewiring.
This study thus represents a confluence of molecular biology, biophysics, and genomics, providing one of the first mechanistic frameworks linking phase separation to the acquisition of developmental totipotency. It challenges existing dogmas that primarily attribute lineage specification to DNA-binding transcription factors in isolation, emphasizing instead the microenvironmental context created by biomolecular condensation. Such insights herald a new chapter in developmental epigenetics, enriching our comprehension of how spatial and biochemical nuclear organization coalesce to govern life’s earliest developmental steps.
Moving forward, these findings provoke compelling questions for future research: How are DUX condensate dynamics regulated temporally and spatially during embryogenesis? What additional molecular components modulate condensate composition and function? Can the principles learned from DUX phase separation be generalized to other master regulators of cell identity? Addressing these will be critical for harnessing phase separation in biomedical applications and elucidating its broader roles in genome biology.
In conclusion, the uncovering of DUX-mediated phase separation as a mechanistic cornerstone of 3D genome reorganization and totipotency gene activation profoundly advances our understanding of early developmental programming. By revealing how dynamic, multivalent protein assemblies orchestrate nuclear architecture and transcription, this research not only solves an enduring biological puzzle but also opens exciting translational opportunities in regenerative medicine and genetic disease intervention.
Subject of Research: Not applicable
Article Title: Phase separation of DUX family proteins drives totipotent-like state via 3D genome reorganization and retrotransposon activation
News Publication Date: 15-Mar-2026
Web References: 10.1093/procel/pwag014
Image Credits: HIGHER EDUCATION PRESS
Keywords: Cell biology, phase separation, totipotency, DUX proteins, 3D genome organization, super-enhancers, retrotransposon activation, zygotic genome activation, chromatin remodeling, biomolecular condensates, transcriptional regulation, developmental epigenetics, Facioscapulohumeral Muscular Dystrophy
Tags: 3D genome remodelingarginine residues role in phase separationbiomolecular condensates in genome organizationchromatin reconfiguration during early developmentDUX family protein phase separationendogenous retrotransposon activationintrinsically disordered regions in DUXliquid-liquid phase separation in transcriptionMERVL expression regulationtotipotent-like state inductiontranscriptional regulation of totipotencyzygotic genome activation mechanisms