In a groundbreaking development that has captured the attention of the neuroscience community, a team of researchers has uncovered a remarkable therapeutic potential for haloperidol, a drug traditionally used as an antipsychotic, in the treatment of spinal muscular atrophy (SMA). The study, published in Experimental & Molecular Medicine on April 13, 2026, reveals that haloperidol not only confers neuroprotection but also significantly improves neuromuscular function in both murine and human models of SMA, marking a major leap forward in the quest for effective treatments against this debilitating genetic disorder.
Spinal muscular atrophy is a devastating neuromuscular condition characterized by the progressive degeneration of motor neurons in the spinal cord, leading to muscle weakness, atrophy, and ultimately paralysis. This autosomal recessive disease primarily results from mutations in the SMN1 gene, which compromise the production of the survival motor neuron (SMN) protein—a crucial factor for motor neuron health and synaptic integrity. Given the complex pathology of SMA, effective treatments have been elusive, and current therapeutic approaches mainly focus on increasing SMN protein levels or symptomatic management rather than providing comprehensive neuroprotection.
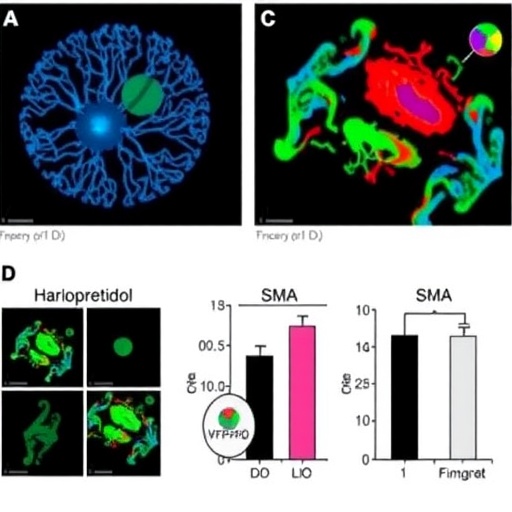
The novel findings around haloperidol suggest an unexpected mechanism for neuroprotection that extends beyond its established role as a dopamine receptor antagonist. The research team employed both murine models genetically engineered to mimic human SMA and induced pluripotent stem cell-derived motor neurons from SMA patients to evaluate the drug’s efficacy. Impressively, haloperidol treatment resulted in marked preservation of motor neurons, enhanced motor function, and overall neuromuscular improvement compared to untreated controls.
Central to the study’s technical breakthroughs was the detailed interrogation of haloperidol’s multipronged effects on cellular pathways implicated in motor neuron survival. Using advanced molecular techniques, the researchers demonstrated that haloperidol modulates neuroinflammatory responses and mitigates oxidative stress—two critical drivers of neuronal degeneration in SMA. Furthermore, haloperidol appeared to facilitate synaptic stabilization by promoting the interaction between motor neurons and their muscle targets, thus preserving functional neuromuscular junctions that are typically compromised in SMA pathology.
In addition to neuroprotection, haloperidol’s enhancement of neuromuscular function was shown through rigorous behavioral and electrophysiological assays in animal models. Treated mice exhibited improved motor coordination, increased grip strength, and prolonged survival, which collectively underscore the drug’s potential to translate into tangible clinical benefits. Similarly, in human cell models, haloperidol reversed SMA-associated deficits in motor neuron firing and synaptic transmission, further corroborating its therapeutic promise.
The application of haloperidol to SMA is particularly compelling given its established pharmacokinetic profile and long-standing clinical use in psychiatric disorders. This repositioning strategy could dramatically accelerate the timeline for clinical translation, bypassing many of the initial safety hurdles typically associated with novel compounds. Nonetheless, the researchers caution that the dosing regimens and potential side effects in the context of SMA require thorough evaluation through carefully designed clinical trials.
The implications of this study extend far beyond SMA. By illuminating previously unrecognized neuroprotective properties of haloperidol, the findings open new avenues for exploring similar pharmacological interventions in other motor neuron diseases, such as amyotrophic lateral sclerosis (ALS), where neurodegeneration and neuromuscular dysfunction are also central pathologies. This work heralds a shift in drug repurposing paradigms, emphasizing comprehensive neurobiological profiling to uncover multifaceted benefits of known drugs.
Mechanistically, the research delineates a complex interplay between haloperidol’s modulation of neurotransmitter systems and intracellular signaling cascades implicated in cell survival and apoptosis. Specifically, the drug was found to attenuate hyperactive microglial inflammation and reduce pro-apoptotic markers, thereby creating a more neuroprotective microenvironment. The use of cutting-edge RNA sequencing further identified upregulation of genes involved in mitochondrial biogenesis and antioxidant defenses, highlighting potential molecular targets for future drug development.
Moreover, the therapeutic effect of haloperidol was not limited to neuronal populations but also encompassed improvements in muscle fiber integrity and metabolic function. Muscle biopsies from treated animals revealed enhanced mitochondrial density and reduced signs of atrophy, suggesting that haloperidol exerts systemic benefits that reinforce motor unit resilience. This holistic approach to treatment could address the multi-organ deficits observed in SMA, advancing a more integrated disease management strategy.
The translational impact of these findings has galvanized efforts toward the initiation of clinical trials aimed at assessing haloperidol’s efficacy and safety in SMA patients. Given the progressive and often fatal nature of SMA, the prospect of a readily available pharmacological option that offers both neuroprotection and functional enhancement generates considerable hope among clinicians and patient communities alike. Patient stratification based on SMA severity and genetic background may also optimize therapeutic outcomes.
This research underscores an important paradigm shift in neurodegenerative disease therapeutics: the repositioning of existing drugs with previously unrecognized biological activities can dramatically shorten the path to effective treatments. The strategic combination of preclinical in vivo and in vitro models employed in this study serves as a robust platform for drug screening and mechanistic elucidation, setting a new standard for translational neuroscience research.
While haloperidol’s neuroprotective capabilities are promising, the study authors emphasize the necessity for cautious optimism. The drug’s known side effects in psychiatric use, including extrapyramidal symptoms and metabolic disturbances, underline the importance of precise dosing regimens tailored specifically for SMA patients. Long-term follow-up and safety profiling will be critical components of forthcoming clinical investigations.
In conclusion, this pioneering study unravels a surprising new role for haloperidol as a neuroprotective agent that significantly enhances neuromuscular function in SMA models. By bridging established psychiatric pharmacology and neurodegenerative disease therapy, the research embodies a transformative approach that could redefine treatment paradigms for SMA and beyond. As the scientific community eagerly awaits clinical trial data, these findings invigorate hope for patients afflicted with this currently incurable disorder.
Subject of Research: Spinal muscular atrophy (SMA) and haloperidol’s therapeutic effects on neuroprotection and neuromuscular function.
Article Title: Haloperidol induces neuroprotection and enhances neuromuscular function in both murine and human models of spinal muscular atrophy.
Article References:
Menduti, G., Perez-Gomez, R., Berenger-Currias, N. et al. Haloperidol induces neuroprotection and enhances neuromuscular function in both murine and human models of spinal muscular atrophy. Exp Mol Med (2026). https://doi.org/10.1038/s12276-026-01689-0
Image Credits: AI Generated
DOI: 13 April 2026
Tags: dopamine receptor antagonist in neuroprotectionexperimental molecular medicine SMA studygenetic neuromuscular disorder treatmentshaloperidol effects on neuromuscular functionhaloperidol impact on motor neuronshaloperidol neuroprotection in spinal muscular atrophyhaloperidol repurposing for SMA therapyneuroprotective drug development for SMASMA muscle weakness and atrophy therapySMA neurodegeneration treatment breakthroughsspinal muscular atrophy SMN1 gene mutationtherapeutic advances in spinal