In the relentless battle against cancer, metastasis remains the most formidable challenge, accounting for approximately 90% of cancer-related deaths. Recent breakthroughs by a research team at Heinrich Heine University Düsseldorf (HHU) have peeled back some of the complexity surrounding how cancer cells break away from primary tumors and establish lethal secondary growths in distant organs. Led by Dr. Tobias Reiff at the Institute of Genetics, this new study, recently published in Nature Communications, leverages the power of Drosophila melanogaster—the common fruit fly—to reveal the molecular choreography that enables cancer cells to navigate tissue boundaries and colonize new environments.
Tumors arise from cells that have acquired mutations capable of subverting normal growth control mechanisms. These rogue cells evade programmed cell death and the immune system’s surveillance, allowing them to proliferate unchecked. While early detection typically facilitates effective interventions like surgery, radiation, and chemotherapy, the insidious spread of cancer cells beyond the primary tumor—metastasis—poses a far deadlier threat. Metastatic cells infiltrate other tissues via blood and lymphatic vessels in a process called dissemination, later homing in on new sites to seed secondary tumors. Understanding the molecular signals guiding this journey is critical to developing therapies that can halt cancer’s spread.
The Düsseldorf team homed in on colorectal cancer, a malignancy often diagnosed late due to subtle symptoms. Dr. Reiff explains that by the time colorectal tumors are detected, cancerous cells might have already embarked on their dissemination journey, complicating treatment options and worsening prognoses. The study focused on deciphering how these cancer cells detach from their primary niche and maneuver through the body, especially how they negotiate organ boundaries—a poorly understood step in metastasis.
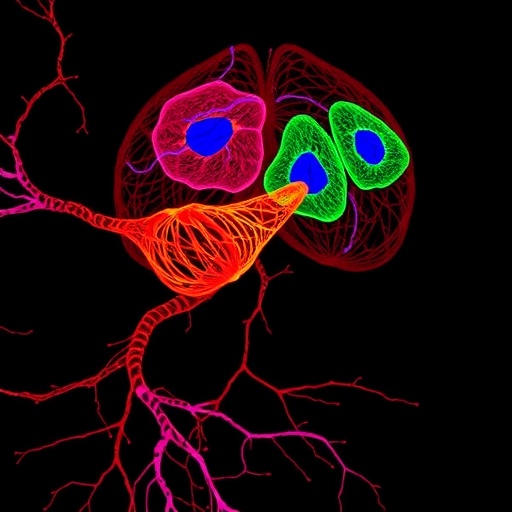
Their investigation unveiled the pivotal role of Netrins, a family of secreted signaling proteins, and their receptor, known as Frazzled/Deleted in Colorectal Cancer (DCC). This receptor-ligand pair orchestrates when and how cancer cells migrate across tissues. Using advanced genetic and live-imaging techniques in Drosophila, the team traced the cellular movements and signaling pathways implicated in this process. The fruit fly model offers the advantage of rapid life cycles, genetic tractability, and conservation of many key oncogenic pathways, making it an invaluable proxy for human cancer research.
Employing state-of-the-art laser microscopy, the researchers tagged intestinal stem cells with fluorescent markers, enabling real-time visualization of their migration patterns within the fly’s gut. This approach, termed the “Hamelin Assay” in homage to the Pied Piper legend, illustrated how Netrins act as chemoattractant signals, guiding the stem cells across the intestinal boundary much like the Piper’s music lured rats away from Hamelin. The analogy elegantly captures the directed movement of cells influenced by precise molecular cues.
Critically, alterations in the DCC receptor gene were found in roughly 65% of colorectal cancer patients, underscoring its clinical significance. These modifications seem to dismantle normal signaling pathways, allowing cancer cells to disengage from the primary tumor and invade neighboring tissues. By elucidating this mechanism, Dr. Reiff’s team has opened a window into potential therapeutic interventions aimed at disrupting Netrin-DCC signaling, thereby preventing early metastasis and improving patient outcomes.
The importance of this discovery lies not only in identifying a new molecular axis controlling metastasis but also in demonstrating the power of model organisms to reflect human disease processes. The comparative genetic architecture between Drosophila and humans—involving conserved pathways regulating cell division, fate, and death—reinforces the relevance of fly-based findings. Such cross-species insights accelerate the translation of fundamental science into clinical applications.
While the study lays crucial groundwork, further research is imperative to fully map the downstream effects of Netrin-DCC signaling in various tissue contexts. Exploring how this axis interfaces with immune evasion, extracellular matrix remodeling, and angiogenesis could yield a holistic understanding of metastatic colonization. Additionally, verifying these molecular interactions in mammalian models and human tissues will be necessary to validate therapeutic targets.
Funding support from the Wilhelm Sander Foundation and Deutsche Krebshilfe enabled the deployment of cutting-edge imaging and genetic manipulation techniques essential to this project. These advances reflect a broader scientific commitment to unraveling cancer’s metastatic puzzle, combining molecular biology, genetics, and live imaging to illuminate processes historically shrouded in obscurity.
In sum, the Nature Communications publication marks a significant step toward intercepting cancer’s deadliest move—metastasis. By charting how Netrin signaling guides intestinal stem cells through organ boundaries, the research team from HHU Düsseldorf provides a promising avenue for therapeutic innovation. The Hamelin Assay’s creative use of the Drosophila model exemplifies how classic biological tools can yield fresh perspectives on contemporary medical challenges, offering hope for better diagnostic markers and treatments against metastatic colorectal cancer.
Subject of Research: Mechanisms underlying metastasis in colorectal cancer and the role of Netrin-DCC signaling in cancer cell migration.
Article Title: Frazzled/DCC directs spatial progenitor integration ensuring steady-state intestinal turnover
News Publication Date: 14-Mar-2026
Web References:
https://www.nature.com/articles/s41467-026-70704-9
References:
Lisa Zipper, Pol Ramon-Cañellas, Filiz Akkas-Gazzoni & Tobias Reiff; Frazzled/DCC directs spatial progenitor integration ensuring steady-state intestinal turnover; Nature Communications 17, 2491 (2026)
Image Credits: HHU/Tobias Reiff
Keywords: Metastasis, Colorectal Cancer, Netrins, DCC Receptor, Frazzled, Drosophila melanogaster, Cancer Cell Migration, Hamelin Assay, Cancer Signaling Pathways, Laser Microscopy, Stem Cell Migration, Cancer Therapy Development
Tags: cancer cell migration and invasioncancer metastasis mechanismsDrosophila models for tumor studygenetic mutations in cancer progressioninnovations in cancer metastasis researchmolecular pathways of cancer spreadmolecular signals in metastasisovercoming cancer cell immune evasionrole of fruit fly in cancer researchsecondary tumor formation biologytargeted therapies for metastatic cancertumor cell dissemination process