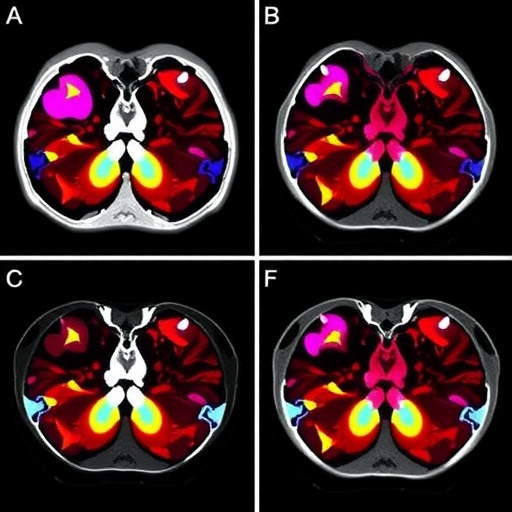
Advancements in the diagnosis and treatment of female-specific cancers such as breast and ovarian cancer have significantly improved patient survival rates over recent years. However, this progress has concurrently unveiled serious long-term complications, the most devastating of which is therapy-related myeloid neoplasms (t-MN). These malignancies, comprising therapy-related acute myeloid leukemia (t-AML) and myelodysplastic syndromes (t-MDS), present a clinical challenge due to their complex pathogenesis and generally poorer prognosis compared to primary myeloid neoplasms. Understanding and mitigating the risk of t-MN is therefore critical not only to prolong survival but also to maintain quality of life in female cancer survivors.
The molecular etiology of t-MN is intricate and multifactorial, stemming from a confluence of treatment-related exposures, genetic predispositions, acquired somatic mutations, and alterations in the hematopoietic microenvironment. Conventional cytotoxic therapies, including alkylating agents and topoisomerase II inhibitors, play a pivotal role by inflicting DNA damage that impairs genomic integrity and functional cellular repair pathways. More recently, targeted agents such as PARP inhibitors have also been implicated in promoting leukemogenesis via mechanisms that compromise DNA repair fidelity. Moreover, radiotherapy contributes additional genotoxic stress, further compounding the risk of clonal hematopoietic aberrations.
Genetic susceptibility critically shapes individual vulnerability to t-MN. Germline mutations in key tumor suppressor genes such as TP53 and BRCA1/2 not only predispose patients to initial malignancies but also exacerbate susceptibility to secondary hematologic neoplasms. These inherited mutations can weaken DNA damage responses, rendering hematopoietic stem and progenitor cells more prone to malignant transformation upon exposure to therapeutic insults. Somatic mutations acquired during or subsequent to therapy act synergistically, promoting clonal expansion and disease progression. Clonal hematopoiesis of indeterminate potential (CHIP) has emerged as a vital harbinger of t-MN, representing an early step in leukemic evolution characterized by the expansion of mutant hematopoietic clones in the absence of overt malignancy.
The bone marrow microenvironment is an often-overlooked arena in t-MN pathophysiology, where aberrant cellular interactions and inflammatory signaling can foster a permissive niche for malignant clones. Alterations in stromal cells, immune components, and extracellular matrix can produce a milieu that favors hematopoietic dysregulation and leukemogenesis, making microenvironmental modulation a potential therapeutic target.
Given the multifaceted origins of t-MN, preemptive risk stratification and personalized treatment planning are paramount. The proposed management framework advocates comprehensive baseline evaluations prior to initiating therapy, encompassing hematologic parameter assessment, organ function testing, exhaustive family history analysis, and advanced genetic profiling to identify germline mutations and somatic aberrations. Integrating CHIP evaluation and epigenetic markers such as DNA methylation status enhances risk prediction precision. These data guide clinicians in selecting treatment regimens that judiciously balance primary cancer eradication with minimizing leukemogenic risk, underscoring the preference for modalities with lower mutagenic potential.
Throughout therapy and in the post-treatment surveillance phase, meticulous monitoring remains essential. Serial blood counts and peripheral smear analyses serve as frontline screens for hematologic abnormalities indicative of emerging t-MN. Detection of abnormal findings should prompt in-depth hematologic evaluations, including chronological exposure documentation to cytotoxic agents and radiotherapy, targeted molecular diagnostics, and timely bone marrow biopsies. Dynamic monitoring of clonal hematopoiesis and mutation profiling facilitates early detection of malignant transformation, enabling prompt clinical intervention.
Therapeutic options for established t-MN continue to pose formidable challenges. Traditional chemotherapeutic approaches often yield suboptimal responses, and hematopoietic stem cell transplantation—considered curative in select cases—is limited by patient eligibility constraints and procedural risks. Nonetheless, evolving treatment paradigms incorporating novel agents bring hope. CPX-351, a liposomal formulation combining cytarabine and daunorubicin, has demonstrated improved efficacy by enhancing drug delivery and minimizing systemic toxicity. Hypomethylating agents, used in conjunction with targeted therapies, are gaining traction for their capacity to reverse aberrant epigenetic modifications driving myeloid malignancies.
Exciting frontiers include immunotherapeutic strategies such as chimeric antigen receptor T-cell (CAR-T) therapy, which harness engineered lymphocytes to specifically eliminate malignant clones. Furthermore, targeting menin, a protein implicated in chromatin regulation and leukemogenesis, with small-molecule inhibitors represents a cutting-edge approach currently under investigation. These innovative treatments promise to transform the management landscape of t-MN, offering tailored, mechanism-based interventions.
The overarching goal in managing female cancer survivors extends beyond cancer control to encompass the prevention of severe complications like t-MN. Achieving this requires a delicate balance that integrates rigorous tumor eradication protocols with judicious minimization of long-term hematologic toxicity. This comprehensive review articulates a systematic framework for the precise prevention, early detection, and effective management of t-MN in female patients. Incorporation of genetic testing, clonal hematopoiesis evaluation, individualized therapeutic strategies, and vigilant longitudinal surveillance are pivotal pillars in this approach.
As research continues to unravel the molecular underpinnings of therapy-related leukemogenesis and novel therapeutic modalities emerge, the prospect of full-process precision management becomes increasingly tangible. Ultimately, these advances hold the promise of constructing a robust long-term health barrier for female cancer survivors, ensuring that gains in cancer survival are not overshadowed by debilitating secondary hematologic neoplasms. The future of oncology is poised to embrace integrated, patient-specific strategies that safeguard overall wellness while conquering primary malignancies, heralding a new era of holistic cancer care.
Subject of Research: Therapy-related myeloid neoplasms in female-specific cancers: pathogenesis, risk stratification, and therapeutic advances
Article Title: Precise Risk Management and Innovative Therapeutics for Therapy-Related Myeloid Neoplasms in Female Cancer Survivors
Web References: http://dx.doi.org/10.1016/j.medp.2026.100124
Image Credits: ©Science China Press
Keywords: therapy-related myeloid neoplasms, t-MN, female-specific cancer, breast cancer, ovarian cancer, clonal hematopoiesis, TP53 mutation, BRCA1/2 mutation, DNA repair, leukemogenesis, CPX-351, hypomethylating agents, CAR-T therapy, menin inhibitors
Tags: genetic predisposition to therapy-related leukemiaimpact of alkylating agents on hematopoiesislong-term complications of ovarian cancer treatmentmolecular mechanisms of therapy-related myelomyelodysplastic syndromes post cancer therapyPARP inhibitors and secondary blood cancersradiotherapy-induced clonal hematopoiesisrole of topoisomerase II inhibitors in leukemogenesistherapy-related acute myeloid leukemia after breast cancertherapy-related myeloid neoplasms in female cancer survivors