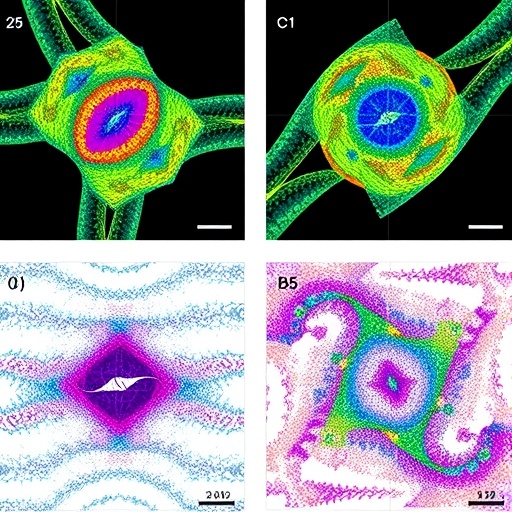
In a groundbreaking advance set to transform our understanding of joint mechanics, a team of interdisciplinary scientists has unveiled a novel imaging technique that probes the micromechanical architecture of joint tissues with unprecedented precision. This innovative method, termed speckle rheological microscopy (SRM), leverages the complex interference patterns—known as speckles—generated by coherent light scattering within biological tissues to reveal detailed biomechanical properties at microscopic scales. The research, published recently in Nature Communications, promises to reshape diagnostic and therapeutic strategies for joint diseases that affect millions worldwide.
Joint tissues such as cartilage, ligaments, and the synovial membrane, possess complex hierarchical structures that confer remarkable mechanical resilience and adaptability. However, traditional imaging modalities have fallen short in capturing the spatial variability of mechanical properties at the microscale. The introduction of SRM addresses this gap by enabling non-invasive, high-resolution mapping of viscoelastic characteristics deep within joint tissues. Unlike conventional rheology techniques that require physical contact or tissue extraction, SRM operates via light scattering phenomena, making it ideal for in vivo applications where preserving tissue integrity is paramount.
At the core of SRM lies the analysis of speckle pattern fluctuations induced by micro-scale mechanical perturbations. When coherent laser light illuminates joint tissue, the scattered photons interfere to create a unique speckle pattern, highly sensitive to the underlying mechanical environment. By monitoring how these speckles decorrelate over time in response to controlled mechanical stimulation, researchers can infer viscoelastic parameters such as stiffness and damping at a spatial resolution down to tens of micrometers. This capability opens new vistas for characterizing the nuanced biomechanical heterogeneity within cartilage layers that dictate functional performance and disease progression.
The research team, led by Leartprapun, Guastaldi, and Randolph, integrated advanced optical instrumentation with sophisticated computational models inspired by rheological theory. Their approach couples speckle pattern acquisition with multifrequency mechanical excitation protocols, enabling the disentanglement of complex time-dependent tissue responses. By applying this method to ex vivo and in situ specimens from animal models, they successfully reconstructed 3D maps of tissue micromechanics, validating the accuracy through complementary histological and mechanical tests. These findings illuminate the intricate interplay between tissue microstructure and mechanical function that has remained obscured until now.
Beyond the methodological innovation, the implications for clinical and biomedical research are profound. Osteoarthritis and other degenerative joint disorders are characterized by subtle changes in tissue stiffness and viscoelasticity that precede gross morphological damage. SRM could thus serve as a sensitive early diagnostic tool, detecting aberrations long before symptoms manifest. Moreover, the fine-scale mechanical mapping informs mechanobiological studies exploring how mechanical forces govern cellular behavior within joints, fostering the development of therapies that target mechanical microenvironments to promote tissue regeneration and repair.
Detailed examination of joint tissue mechanical heterogeneity by SRM also advances implant design and biomaterial engineering. Prosthetics and scaffolds must mimic native tissue mechanics to ensure integration and durability. The rich datasets generated by speckle rheological microscopy provide foundational knowledge to customize material properties at microscale, potentially enhancing implant longevity and patient outcomes. This bridges a longstanding gap between biomechanical theory and translational applications, enabling a new era of personalized joint medicine.
Technical refinements implemented in this study address several challenges that have historically limited speckle-based imaging in tissues. The authors employed adaptive optics techniques to mitigate signal degradation caused by tissue scattering and motion artifacts, thereby enhancing resolution and repeatability. Additionally, advanced statistical algorithms enable robust extraction of mechanical parameters from inherently noisy speckle signals, paving the way for automated, high-throughput analysis pipelines. Such technological sophistication ensures the method’s scalability for large cohort studies and eventual clinical deployment.
A particularly striking aspect of the research is the multidimensional insight gained by correlating micromechanical maps with immunohistochemical markers of tissue composition and inflammation. This comprehensive approach reveals how mechanical alterations co-localize with biochemical changes, offering a holistic perspective on the pathophysiology of joint disorders. The interplay between mechanics and molecular environment underscores the complexity of joint tissue homeostasis and highlights the necessity of integrated diagnostic platforms like SRM.
The study also explored how dynamic loading conditions influence speckle pattern decorrelation, simulating physiological joint movements. These experiments demonstrate that SRM can capture time-dependent viscoelastic responses under cyclic stress, reflecting real-world mechanical environments. This dynamic assessment capability marks a departure from static measurements, providing richer information to understand tissue adaptation, fatigue, and failure mechanisms—critical for designing prevention strategies for injury and degeneration.
Importantly, the researchers validated their SRM measurements across multiple joint tissues, including articular cartilage, meniscus, and ligamentous structures. This versatility indicates broad applicability across musculoskeletal research fields. Furthermore, the non-destructive nature of the technique allows longitudinal monitoring in preclinical models, facilitating studies of tissue development, aging, and response to interventions over time. Such longitudinal data sets are invaluable for deciphering temporal sequences that underlie joint health and disease trajectories.
The potential translation of SRM into clinical settings hinges on developing compact, user-friendly instrumentation compatible with arthroscopic tools or handheld devices. The authors outline preliminary designs incorporating fiber optic probes and miniaturized laser sources, signaling a roadmap toward real-time, minimally invasive joint assessments. Integrating SRM with existing imaging platforms such as MRI or ultrasound could yield multimodal diagnostics that simultaneously capture structural, biochemical, and mechanical information—ushering in unprecedented diagnostic precision.
As the scientific community grapples with growing burdens of musculoskeletal diseases driven by aging populations and lifestyle factors, this research offers timely solutions grounded in physics and engineering innovations. The fusion of optical physics, rheology, and biomechanics represented by speckle rheological microscopy exemplifies the interdisciplinary approaches required to tackle complex biological problems. By providing deeper understanding of joint microenvironment mechanics, it sets a new standard for basic and translational research aimed at alleviating joint dysfunction globally.
Future directions envisioned by the team include expanding SRM to study soft tissues beyond joints, such as intervertebral discs, tendons, and even brain tissue, where mechanical properties critically influence function and pathology. The modularity of the technique promises adaptability to diverse tissue types, fostering broad biomedical and clinical applications. Moreover, integrating machine learning algorithms to analyze complex speckle datasets stands to enhance diagnostic accuracy and reveal subtle patterns beyond human detection, propelling SRM into the era of smart medical diagnostics.
In summary, the development of speckle rheological microscopy represents a transformative leap in our capacity to visualize and quantify the micromechanical landscapes of joint tissues. By marrying optical speckle analysis with rheological frameworks, this method uncovers the delicate balance of forces sustaining joint integrity and function. The work of Leartprapun, Guastaldi, Randolph, and colleagues illuminates new frontiers in musculoskeletal science, with far-reaching implications for disease diagnosis, treatment, and prevention. As this technology evolves, it holds immense promise to revolutionize personalized joint care and improve the quality of life for millions affected by joint ailments worldwide.
Subject of Research: Assessing the micromechanical architecture of joint tissues
Article Title: Assessing the micromechanical architecture of joint tissues with speckle rheological microscopy
Article References:
Leartprapun, N., Guastaldi, F.P.S., Randolph, M.A. et al. Assessing the micromechanical architecture of joint tissues with speckle rheological microscopy. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70248-y
Image Credits: AI Generated
Tags: advanced imaging for joint diseasesbiomechanical properties of cartilagecoherent light scattering in biologyhigh-resolution rheology techniquesin vivo joint tissue analysisjoint tissue micromechanicsmicromechanical architecture of jointsnon-invasive joint imagingspeckle pattern fluctuation analysisspeckle rheological microscopysynovial membrane biomechanicsviscoelastic mapping of ligaments