Credit: Image created by Ada H. V. Repetto-Llamazares, Nordic Nanovector ASA.
Reston, VA–A new radioimmunotherapy has proven effective in reversing resistance to the most commonly used lymphoma drug, rituximab, according to research published in the October issue of The Journal of Nuclear Medicine. When used in combination with rituximab, 177Lu-lilotomab-satetraxetan was shown to substantially increase rituximab binding and rituximab-mediated antibody-dependent cellular cytotoxicity (ADCC) activity, resulting in significant tumor growth delay in a non-Hodgkin’s lymphoma mouse model.
Non-Hodgkin’s lymphoma is the most common blood cancer, with the eleventh highest mortality rate of all malignancies worldwide in 2018, according to the American Cancer Society. The monoclonal antibody rituximab was approved for treatment of non-Hodgkin’s lymphoma more than 20 years ago and is currently the standard of care. However, many patients eventually develop resistance against rituximab, which is often associated with changes in expression of the CD20 antigen.
177Lu-lilotomab-satetraxetan (177Lu-lilotomab)–a next-generation b-particle-emitting radioimmunoconjugate–has been shown to increase CD20 expression in different rituximab-sensitive non-Hodgkin’s lymphoma cell lines and to act synergistically with rituximab in a non-Hodgkin’s lymphoma animal model. As such, researchers hypothesized that 177Lu-lilotomab could be used to reverse rituximab resistance in non-Hodgkin’s lymphoma.
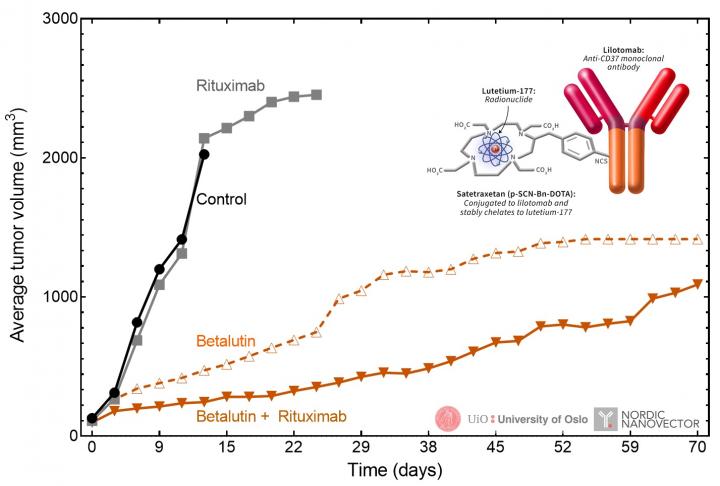
In the study, two non-Hodgkin’s lymphoma cell lines–Raji (parent line) and Raji2R (rituximab-resistant line)–were cultured and incubated with either lilotomab, 177Lu-lilotomab or saline. Xenografted mice were then administered either saline, rituximab monotherapy, 177Lu-lilotomab monotherapy or a combination therapy of 177Lu-lilotomab-satetraxetan and rituximab. Tumor volume and survival time were calculated and analyzed.
Exposure of the cell lines to 177Lu-lilotomab resulted in an increase in rituximab binding, as compared with control cells. With no 177Lu-lilotomab exposure, binding in the rituximab-resistant Raji2R cells was on average 36±5 percent compared to untreated Raji cells. After treatment with 177Lu-lilotomab, the rituximab-binding in Raji2R cells increased to 53±3 percent. In contrast, treatment with unlabeled lilotomab or saline had no effect on rituximab binding. Treatment with 177Lu-lilotomab also increased ADCC induction to 30±3 percent of Raji cells, representing a 50 percent increase.
In the xenografted mice, the combination of rituximab with 177Lu-lilotomab synergistically suppressed RajiR2 tumor growth. The median survival time of mice treated with this combination doubled when compared to survival of mice given 177Lu-lilotomab monotherapy and was five times longer than for mice given rituximab monotherapy.
“This work is potentially very important, as it could be a last way out for patients that have become resistant to rituximab. If those patients receive an injection of 177Lu-lilotomab-satetraxetan, they can be treated again with rituximab and have an improved effect,” said Dr. Jostein Dahle, PhD, chief scientific officer at Nordic Nanovector. “In a phase 1b clinical trial, a 100 percent complete response rate was achieved in the first group of patients treated with 177Lu-lilotomab-satetraxetan followed by rituximab. Achieving a complete response is very important, since it usually correlates with an improved duration of response and overall survival.”
Dahle continued, “Combination treatments are the future for cancer therapy. By exploring strategies with radioimmunotherapy together with other drugs, nuclear medicine may play an important role in lymphoma therapy.”
###
The authors of “177Lu-Lilotomab Satetraxetan has the Potential to Counteract Resistance to Rituximab in Non-Hodgkin’s Lymphoma” include Marion M. Malenge, Nordic Nanovector ASA, Oslo, Norway, Department of Radiation Biology, Institute for Cancer Research, Oslo University Hospital, Oslo, Norway, and Institute for Clinical Medicine, University of Oslo, Oslo, Norway; Sebastian Patzke, Nordic Nanovector ASA, Oslo, Norway, and Department of Radiation Biology, Institute for Cancer Research, Oslo University Hospital, Oslo, Norway; Anne H. Ree, Institute for Clinical Medicine, University of Oslo, Oslo, Norway, and Department of Oncology, Akershus University Hospital, Norway; Trond Stokke, Department of Radiation Biology, Institute for Cancer Research, Oslo University Hospital, Oslo, Norway; Peter Ceuppens and Brian Middleton, Inferstats Consulting Ltd., Cheshire, United Kingdom; and Jostein Dahle and Ada H. V. Repetto-Llamazares, Nordic Nanovector ASA, Oslo, Norway.
This study was made available online in April 2020 ahead of final publication in print in October 2020.
Please visit the SNMMI Media Center for more information about molecular imaging and precision imaging. To schedule an interview with the researchers, please contact Rebecca Maxey at (703) 652-6772 or [email protected].
About the Society of Nuclear Medicine and Molecular Imaging
The Journal of Nuclear Medicine (JNM) is the world’s leading nuclear medicine, molecular imaging and theranostics journal, accessed close to 10 million times each year by practitioners around the globe, providing them with the information they need to advance this rapidly expanding field. Current and past issues of The Journal of Nuclear Medicine can be found online at http://jnm.
JNM is published by the Society of Nuclear Medicine and Molecular Imaging (SNMMI), an international scientific and medical organization dedicated to advancing nuclear medicine and molecular imaging–precision medicine that allows diagnosis and treatment to be tailored to individual patients in order to achieve the best possible outcomes. For more information, visit http://www.
Media Contact
Rebecca Maxey
[email protected]
Original Source
http://www.
Related Journal Article
http://dx.



