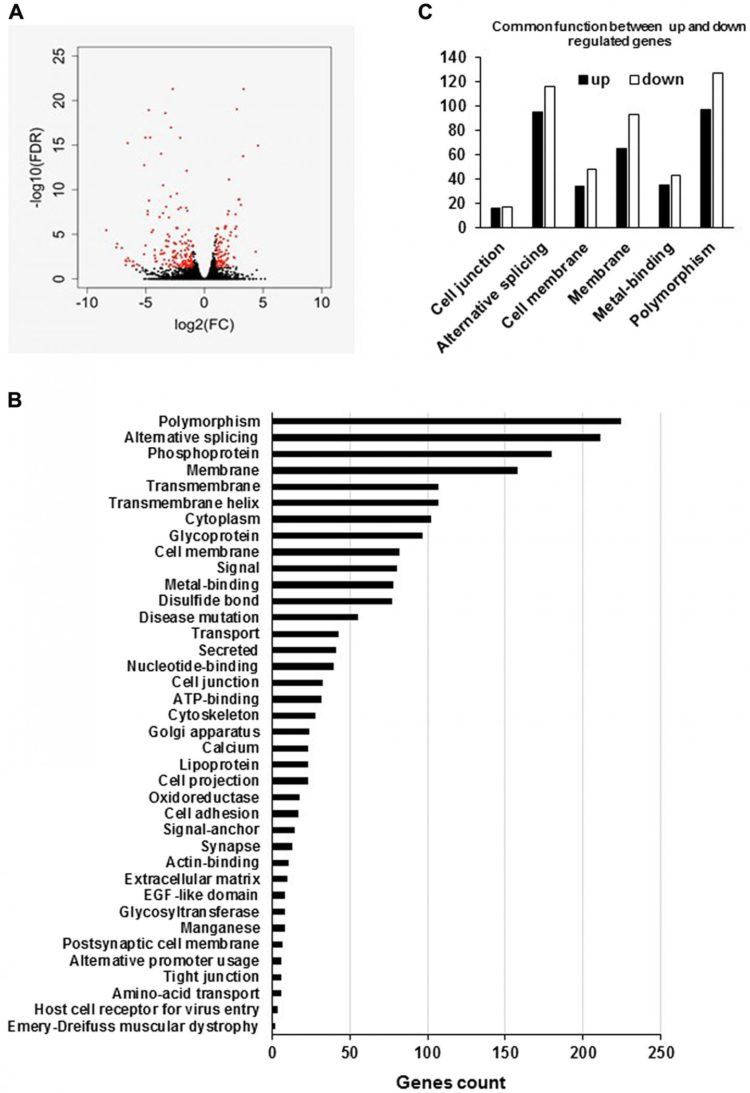
The cover for issue 12 of Oncotarget features Figure 11, ‘Global analyses of the RNA-Seq data of LNCaP empty vector and LNCaP cells overexpressing hPCL3S (Clone 12)’ by Abdelfettah, et al.
Credit: Correspondence to – Dominique Leprince – [email protected]
The cover for issue 12 of Oncotarget features Figure 11, “Global analyses of the RNA-Seq data of LNCaP empty vector and LNCaP cells overexpressing hPCL3S (Clone 12)” by Abdelfettah, et al.
These proteins contain a TUDOR domain binding H3K36me3, two PHD domains and a Winged-helix domain involved in GC-rich DNA binding.
The human PCL3 locus encodes the full-length hPCL3L protein and a shorter isoform, hPCL3S containing the TUDOR and PHD1 domains only.
A mutant hPCL3S unable to bind H3K36me3 increased proliferation and migration of LNCa P similarly to wt hPCL3S whereas inactivation of its PHD1 domain decreased proliferation.
Dr. Dominique Leprince from the University de Lille, CNRS, Institut Pasteur de Lille, UMR 8161m M3T, Mechanisms of Tumorigenesis and Targeted Therapies, F-59000 Lille, France said, “Prostate cancer (PCa) is the second most common type of cancer diagnosed worldwide and is still a leading cause of death despite recent research advances.“
“Prostate cancer (PCa) is the second most common type of cancer diagnosed worldwide and is still a leading cause of death despite recent research advances”
– Dr. Dominique Leprince, University de Lille, CNRS, Institut Pasteur de Lille, UMR 8161m M3T, Mechanisms of Tumorigenesis and Targeted Therapies
Two of most commonly used prostate cancer cell lines LNCa P and PC3 are representative of prostatic adenocarcinoma and of Small Cell Neuroendocrine Carcinomas, respectively.
These proteins share a structured N-terminal domain consisting of a TUDOR domain, two PHD domains followed by a Winged-helix domain involved in GC-rich DNA binding and a C-terminal reverse Chromodomain.
Indeed, PHF1 through its PHD1 domain is the only human Polycomb-like protein capable of inducing cell quiescence by interacting with P53 to stabilize it independently of its TUDOR domain and thus of its binding at chromatin.
In this study, the authors have quantified the expression levels of both hPCL3 isoforms in primary prostate tumors as well as in the hormone-dependent LNCa P and hormone-independent DU145 and PC3 prostate cancer cell lines.
Collectively, their results provide insights into a new mechanism whereby AR-independent prostate cancer cell lines acquire heightened ability to proliferate and migrate and highlight hPCL3S targeting as a new potential interventional strategy against castration resistant prostate cancers.
The Leprince Research Team concluded in their Oncotarget Research Paper, “we demonstrated that overexpression of hPCL3S promoted proliferation, anchorage-independent growth and migration in human LNCaP prostate cancer cells whereas silencing of endogenous hPCL3S in DU145 and PC3 prostate cancer cells impaired these effects. Furthermore, our results suggested that hPCL3S did not act through the perturbation of PRC2 activity but rather mainly through chromatin-independent effects relying on its PHD1 domain. Therefore, our studies could have identified the overexpression of hPCL3S as a new alteration contributing to tumor progression in prostate cancer through the emergence of a rapidly growing cell population of neuroendocrine-like cells.“
###
Sign up for free Altmetric alerts about this article
DOI – https:/
Full text – https:/
Correspondence to – Dominique Leprince – [email protected]
Keywords –
hPCL3S,
PHF19,
PRC2,
β-catenin,
prostate cancer
About Oncotarget
Oncotarget is a weekly, peer-reviewed, open access biomedical journal covering research on all aspects of oncology.
To learn more about Oncotarget, please visit https:/
SoundCloud – https:/
Facebook – https:/
Twitter – https:/
LinkedIn – https:/
Pinterest – https:/
Reddit – https:/
Oncotarget is published by Impact Journals, LLC please visit http://www.
Media Contact
[email protected]
18009220957×105
Media Contact
Ryan James Jessup
[email protected]
202-638-9720
Original Source
https:/
Related Journal Article
http://dx.