Credit: Kasper Røjkjær Andersen
New Danish research provides mechanistic insight into how DNA is monitored and repaired if damage occurs. The results may eventually help to improve the treatment of certain types of cancer, as the DNA repair complex provides a mechanism for cancer cells to resist chemotherapy.
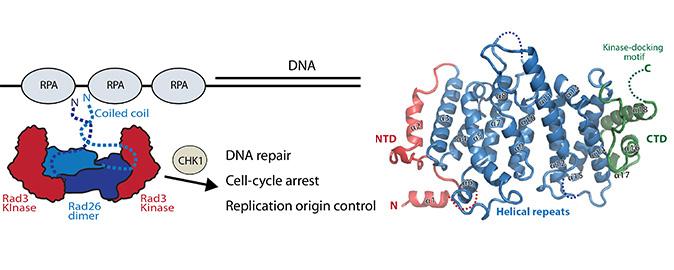
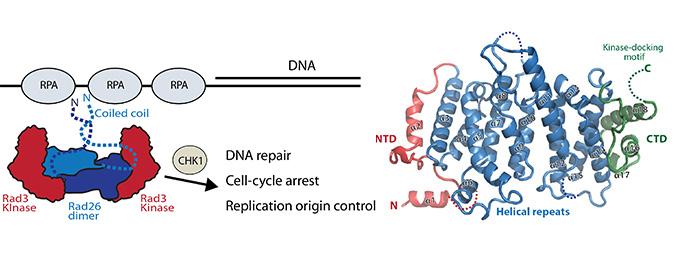
Our DNA is constantly exposed to damage and to protect the genome, cells have evolved mechanisms that monitor and repair these damages. Our structural knowledge of the protein complexes that monitor DNA has so far been limited. New research now describes the structure and organisation of the DNA controls protein Rad26 and shows how the kinase Rad3 is recruited to sites of damaged DNA.
To maintain the genome integrity, DNA damages have to be monitored and repaired. The first step in this process is orchestrated by the Rad3 kinase. Rad26 is a functional subunit of Rad3-rad26 DNA repair complex and is responsible for bringing the kinase to sites of DNA damage, but the mechanism behind kinase recruitment and structural knowledge of how this complete is organized has until now been unclear.
New results from Aarhus reveal the crystal structure of Rad26 and identify the elements that are important for recruiting Rad3 kinase. Rad26 is a dimer with a conserved interface in the N-terminal part of the protein. Biochemical data demonstrated that Rad26 uses its C-terminal domain and a conserved motif to recruit Rad3. From the in vitro reconstituted Rad3-Rad26 complex, small-angle X-ray scattering and electron microscopic studies, it is possible to model the quaternary structure and thus bring us closer to a mechanistic understanding Rad3-Rad26 DNA repair apparatus.
Rad3 signalling ensures that cells do not divide before DNA damages are repaired and thus provides cancer cells with a mechanism to resist chemotherapy by repairing these DNA damages. Our new structural knowledge will help the development of Rad3 inhibitors that make cancer cells more susceptible to chemotherapy and this new treatment is now being tested in clinical trials.
###
The results were published in The Journal of Biological Chemistry: doi: 10.1074/jbc.M117.780189.
Media Contact
Kasper Røjkjær Andersen
[email protected]
45-87-15-49-28
@aarhusuni
http://www.au.dk
############
Story Source: Materials provided by Scienmag